Concentration Units Continued
description
Transcript of Concentration Units Continued

Concentration Units Continued
M =moles of solute
liters of solution
Molarity (M)
Molality (m)
m =moles of solute
mass of solvent (kg)
12.3

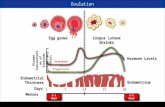
Solution StoichiometryThe concentration of a solution is the amount of solute present in a given quantity of solvent or solution.
M = molarity =moles of solute
liters of solution
How many moles of KI are required to make 500. mL of a 2.80 M KI solution?

Solution StoichiometryThe concentration of a solution is the amount of solute present in a given quantity of solvent or solution. Another way this can be expressed is in mass percentage.
Mass %=mass of solute
mass of solution
What is the mass percentage of 2 moles of NaOH dissolved in 358 ml of water?
X 100

Solution StoichiometryThe concentration of a solution can also be expressed as the moles of solute per kg of solvent.
m = molality =moles of solute
kg of solvent
What is the molality of a solution that has contains83.05g of KI dissolved in 500ml of water?

What is the molality of a solution made from 155g of sodium chloride and 1500g of water?
m =moles of solute
mass of solvent (kg)155g of NaCl = 2.65 mol NaCl 1500g of water = 1.5kg of water
____________________2.65 moles NaCl
1.5kg water = 1.8m

Change in Boiling Point Common Applications of
Boiling Point Elevation

12.6

What is the freezing point of a solution containing 478 g of ethylene glycol (antifreeze) in 3202 g of water? The molar mass of ethylene glycol is 62.01 g.
DTf = Kf m
m =moles of solute
mass of solvent (kg)= 2.41 m=
3.202 kg solvent
478 g x 1 mol62.01 g
Kf water = 1.86 0C/m
DTf = Kf m = 1.86 0C/m x 2.41 m = 4.48 0C
DTf = T f – Tf0
Tf = T f – DTf0 = 0.00 0C – 4.48 0C = -4.48 0C
12.6

Colligative Properties of Electrolyte Solutions
12.7
0.1 m NaCl solution 0.1 m Na+ ions & 0.1 m Cl- ions
Colligative properties are properties that depend only on the number of solute particles in solution and not on the nature of the solute particles.
0.1 m NaCl solution 0.2 m ions in solution
van’t Hoff factor (i) = actual number of particles in soln after dissociation
number of formula units initially dissolved in soln
nonelectrolytesNaClCaCl2
i should be
123

Which chemical below would be bestto de-ice a frozen street and why?
a) sand, SiO2
b) Rock salt, NaClc) Ice Melt, CaCl2
Change in Freezing Point

At what temperature will a 5.4 molal solution of NaCl freeze?
Solution
∆TFP = Kf • m • i
∆TFP = (1.86 oC/molal) • 5.4 m • 2
∆TFP = 20. oC
FP = 0 – 20. = -20. oC
Freezing Point Depression

Triple point diagram
solid
liquid
gas

Phase diagram