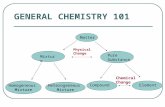
Classifying Matter 8/26. Mixture vs. Pure Substance Matter Pure Substance ElementCompound Mixture...
-
Upload
hortense-oconnor -
Category
Documents
-
view
232 -
download
0
Transcript of Classifying Matter 8/26. Mixture vs. Pure Substance Matter Pure Substance ElementCompound Mixture...
Pure Substances
• Pure Substance- Constant and uniform throughout (atom or molecule) 1 type of somethingAtom Molecule
• Atom- Single element (Cannot be broken into smaller parts)– Elements (ex. Au, H, Ne)– Where can you find a list of elements?
• HOFBrINCl
• Molecule- 2 or more atoms put together
Molecules/Compounds
• Compound- more than one type of element in a molecule (ex. NaCl)
• Can be separated into elements
• The properties of compounds can be much different than the properties of just the elements– For example : NaCl
+
• These elements combine with each other during a chemical reaction
• They combine in a fixed proportion (Law of Definite Proportion)-they bind the same way every time! – Examples – NaCl, H2O, Fe(NO3)3
+
Fun Fact
• Today, there are approximately 10 million known compounds, and new compounds continue to be developed and discovered at the rate of about 100 000 per year.
• There appears to be no limit to the number of compounds that can be made or that will be discovered.
Allotrope
• Allotrope-forms of an element in the same physical state but have different structures–Example: Diamonds and Graphite
(forms of Solid Carbon)
Mixtures
• Mixture- A blend or combination of two or more pure substances
• Not chemically combined!!!!!• Variable compositions (not uniform and unchanging)
– Granite -Candy
Heterogeneous Mixture
• heterogeneous mixture – you can actually see a difference / does not blend smoothly throughout–Examples: Sand & Water, Salt &
Pepper, a bag of M&Ms
Homogeneous Mixture
• homogeneous mixture - you cannot see a difference; it always has a single phase–Examples: Salt water: H2O + NaCl
Air: N2 + O2 + CO2
• Mixture with no visibly different parts
Alloy
• Alloy- a homogeneous mixture of metals or a mixture of a metal and a nonmetal in which the metal substance is the major component– Examples: Steel, Brass
Classify
• Mixtures– Homogeneous– Heterogeneous
• pure substances– elements– compounds
Draw a picture, similar to the pictures above, that represents a compound
Practice
• http://www.teacherbridge.org/public/bhs/teachers/Dana/Matter.html
Create a Matter Concept Map
• Work with your partner to create your very own concept map. (Bubble map)– Be Creative– Use Color– Use Pictures