Classification of Matter Chapter 2. Objectives LWBAT: 1.Describe the characteristics of physical and...
-
Upload
randall-benson -
Category
Documents
-
view
224 -
download
0
Transcript of Classification of Matter Chapter 2. Objectives LWBAT: 1.Describe the characteristics of physical and...

Classification of Matter
Chapter 2

Objectives
LWBAT:
1.Describe the characteristics of physical and chemical properties.
2.Classify mixtures as homogenous or heterogeneous.
3.Describe the characteristics of physical and chemical changes.

What is matter?
Matter is anything that has mass and takes up space.

MaterialsSpecific types of matter
Described according to phases (solid, liquid, gas)

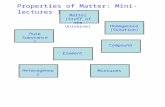
Pure SubstancesContain only one type of matterCannot be separated physicallyHas a fixed composition (structure)

Pure Substances
Elements - simplest pure substances made of only one type of atom (Periodic Table)Cannot be broken down into a simpler substances.

Pure Substances
Compounds - – two or more elements chemically combined (H2O, CuSO4)
Always in a fixed proportion (relationship)

MixturesPhysical combination of two or more substances
Can be separated physically

Heterogeneous MixturesPrefix “Hetero-” means differentSubstances can be easily distinguished and separated
Ex: sand and salt, trail mix, sand and gravel, salad

Homogeneous Mixtures
Prefix “Homo-” means same
Substances are well mixed and appear the same throughout
Ex: Solution

SolutionsSolute dissolves in solvent to form solution.
(solute + solvent = solution)Concentrated solution contains more solute than dilute solution.
Ex: Kool-Aid, iced tea, coffee

Physical Properties(appearance changes but materials
stays the same)
Describe substances undergoing physical changes
Ex: viscosity, conductivity, malleability, hardness, density, boiling point and melting point

ViscosityThe tendency of a liquid to keep from flowing—its resistance to flowing. Example:
Honey – high viscosity Vinegar – low viscosity

Conductivity
A material’s ability to allow heat to flow
High conductivity – metal spoon Low conductivity – wooden spoon

Malleability
Ability of a solid to be hammered without shattering

Hardness
One way to compare the hardness of two materials is to see which of the materials can scratch the other.

DensityCan be used to test the purity of a substanceFormula: mass/volume
Example: testing fuel in motorcycles

Melting and Boiling Points
Melting point – temperature at which a substance changes from a solid to a liquidBoiling point – temperature in which a substance boils (liquid to a gas)

Additional Physical Properties
Color Shape (structure)Texture (how the material feels)

Using Properties to Separate Mixtures
Filtration – a process that separates materials based on the size of the particles.Distillation – a process that separates the substances in a solution based on their boiling point.

Chemical Properties
Describe substances undergoing chemical changes
Ex: flammability, reactivity

Flammability
A material’s ability to burn in the presence of oxygen.

Reactivity
The property that describes how a substance combines chemically with other substances; forms a new substance
Iron + water = rust

Is it a chemical or physical change?
When matter undergoes a chemical change, the composition of the matter changes. When matter undergoes a physical change, the composition of the matter remains the same.

Law of Conservation of Matter
Matter is not created or destroyed during a physical or chemical change
Physical Change Chemical Change

Physical ChangesSame substance remains after changeUsed to separate mixtures
Ex: pounding, tearing, cutting, dissolving, evaporating, melting, boiling, pulling

Chemical ChangesResults in new substance with new propertiesAlso known as a chemical reaction
Ex: burning, digesting, fermenting, decomposing

Chemical Change Evidence1. Precipitate forms (solid)2. Gas evolves (bubbles)3. Color change (tarnishing)4. Energy change
•Exothermic = releases energy•Endothermic = absorbs energy
5. Odor produced (sometimes)