Chemistry Jeopardy Chpt. 3
description
Transcript of Chemistry Jeopardy Chpt. 3

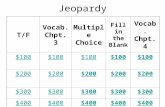
Chemistry Jeopardy Chpt. 3Law Dogs It’s an Atom An Atom a
DaySet the
Periodic Table
How Trendy
10 10 10 10 10
20 20 20 20 20
30 30 30 30 30
40 40 40 40 40
50 50 50 50 50

This law can be used to explain
why it is possible that CO in
Denmark is the same as CO in
Newfoundland.
A. Conservation of mass
B. Definite Proportions
C. Daltons Atomic Theory
D. Law of Multiple Proportions

Because of this law matter is not created or destroyed.
A. Conservation of massB. Definite ProportionsC. Daltons Atomic TheoryD. Law of Multiple Proportions

This scientist proposed the first atomic theory based on
observed laws.
A. RutherfordB. JJ ThomsonC. Einstein D. Dalton

4.00 grams of hydrogen reacts with 32.00 grams of oxygen to form this many
grams of water.
A. 4.00 gB. 16.00 gC. 28.00 gD. 36.00 g

This father of the modern periodic table said that properties of
elements will recur in periodically when arranged by increasing
atomic mass.
A. MillikenB. BohrC. MendeleevD. Boyle

Subatomic particle associated with cathode rays, JJ Thomson, and
negative charge.
A. QuarkB. ElectronC. ProtonD. Neutron

Experimenter that proposed that an atom has a nucleus and is mostly empty space.
A. RutherfordB. ThomsonC. CurieD. Newton

Experiment used in the discovery of the nucleus.
A. Cathode Ray TubeB. Oil Drop ExperimentC. Gold Foil ExperimentD. Manhattan Project

Type of “bullets” fired at gold foil.
A. Alpha ParticlesB. 30-06C. ElectronsD. Laser Beams

Model of the atom proposed by JJ Thomson.
A. QuantumB. Plum PuddingC. OrbitalD. Nuclear

It is the atomic number of Barium.
A. 56B. 137C. 195D. 0

What is the mass number for lead-209?
A. 82B. 127C. 207D. 209

Which of the following pairs are isotopes?
A.Ca+2 and Mg+2
B.199Hg and 200Hg C.18O and 19FD.238U and 238Np

Which element, isotope, or ion listed below contains 35 protons, 46 neutrons, and 36 electrons?
A.81Kr+
B.81Br C.81Kr D.81Br –

1/12 the mass of this isotope is defined as 1 amu.
A. Sodium-24B. Chlorine-35C. Uranium-238D. Carbon-12

Areas of similar chemical properties.
A. PeriodsB. GroupsC. BlocksD. Breaks

Group 17
A. HalogensB. Alkali MetalsC. Noble GasesD. Alkali Earth Metals

A main group element
A. LeadB. UraniumC. IronD. Zinc

Element residing at the intersection of group 6 period 4.
A. HfB. SeC. CrD. Ge

Group of the periodic table shown here
A. Alkali MetalsB. HalogensC. ChalcogensD. Noble Gases

Energy required to remove an electron.
A. Electron AffinityB. Ionization EnergyC. ElectronegativityD. Enthalpy

Atom with smallest atomic radius on the periodic table
A. HeB. HC. RnD. Fr

Atom with the highest ionization energy.
A.Fr
B.H
C.Rn
D.He

The first ionization energy of Aluminum is 6eV. The second ionization energy is
A.Less than 6eVB.More than 6eVC.Equal to 6eVD.Cannot be determined

Arrange the following atoms in order of INCREASING size
Sr, Ba, I
A. Sr, Ba, IB. Ba, I, SrC. I, Sr, BaD. Ba, Sr, I