Chemical Cross-linking Coupled With Mass...
Transcript of Chemical Cross-linking Coupled With Mass...

Chemical Cross-linking Coupled With Mass Spectrometry
Dr. Boris [email protected]
05/15/2017
IDeA National Resource for Proteomics
UAMS, Little Rock, Arkansas

Cross-linked proteins and peptides: overview of analytical problems and solutions
Naturally occurring cross-links
• -S-S- bonds• Lys-to-Lys oxidative cross-links• Transglutaminase fusions• Amino- to carboxy- cross-links• Cyclic peptides
Applications of chemical cross-linking
•Protein-protein interactions•Protein-nucleic acid interactions•Cross-linking as a structure determination technique•Cross-linker selection: types of cross-linkers•Enrichment for cross-linked species•Bioinformatics approaches to cross-linked peptide analysis•Examples of cross-linking proteomics studies
Survey of the field status and perspectives
Chemical Cross-linking Coupled With Mass Spectrometry(CXMS)

X
X
X
Inter-peptide(type 2)
Intra- peptide(Type 1)(special case: cyclic peptides)
Dead end(Type 0)
Different Types of Cross-Linked Products
Problem I:
How to distinguish between these products,for a given X ?
Problem II:
What can we do when more than two peptides are cross-linked?
XX
Question: discuss, how cross-linked peptides will differ in their properties from regular, linear peptides
α
β
Type 2,2

Analytical Questions Asked in CXMS
Protein level:
•What are the identities of interacting proteins?
•What are the protein-protein contacts/interface?
•What can we say about architecture of multi-protein complexes using cross-linking?
•How can we constrain protein structure using cross-linking?
Peptide level:
•What are the sequences of cross-linked peptides?
•Where exactly is the cross-link located?
•Is it inter- or intra- peptide cross-link?

Cross-Links in Nature: Disulfide Bonds
Question: discuss occurrence, properties, and function of disulfide bonds in proteins (~20% of all proteins have –S-S-)
What is approximate value of C-S-S-C dihedral angle?
A protein X has 8 cysteins. How many different protein forms are possible (that have different disulfide bond arrangement). Which form has the highest entropy?
OMNITROPE®

Cross-Links in Nature: Disulfide Bonds
Classical example of the folding of Ribonuclease A

Disulfide Bonds: Proteomics Strategies
Problem: -S-S- reduces backbone cleavage efficiency in a MS/MS experiment using electron capture dissociation/electron transfer dissociation (ECD/ETD)Explain why
Top-down approach
Use high resolution mass-spectrometry. Use electron capture dissociation (ECD) along with conventional CIDCovalent and non-covalent interactions complicate top-down analysisIt is advantageous to reduce –S-S- bondsdiscuss possible approaches using –S-S- focused chemistryZhang et al: proteins undergo electrolytic reduction in an electrochemical cell

Conclusion:
Direct top-down mapping of –S-S-bonds is difficult-S-S- need to be reduced prior to the analysis for better sequence %

Disulfide Bonds: Bottom-Up
1. Better suited for large-scale/automation2. Smaller fragments behave better than intact proteins3. Multiple proteases can be used, if needed4. MS3 can solve issues related to poor fragmentation efficiency
Challenges:
1. Data interpretation for large-scale experiment2. Optimization of chromatography and MS acquisition

Disulfide Bonds: Bottom-Up
Nili et al 2012

Disulfide Mapping
ETD cleaves –S-S-preferentially
CID is used for the site assignment
*
Nili et al 2012

Disulfide Mapping: 4 peptides linked
Nili et al 2012

Disulfide Mapping: Data Acquisition and Analysis
Nili et al 2012
C18 chromatography: 90 min 0-30% acetonitrileInstrument: LTQ-Orbitrap Velos with ETDAcquisition: survey MS scan 7 data-dependant scans consisting of collision-induced-dissociation (CID) and ETD MS2 scans on the most intense ion in the survey scan, followed by 5 MS3 CID scans on the 1st to 5th most intense ions in the ETD MS2 scan.Inclusion lists for cross-linked species (follow-up experiments)Data analysis:Bioworks: Charge state analysis. Prepare separate peak lists for MS2 and MS3 data; and search each with 2.5 Da parent mass tolerance, and 1 Da fragment tol.Final analysis: Scaffold

Cyclic Peptides
Journal of Proteome Research 2011

Cyclic Peptides

Cycloquest: Search Algorithm for cyclic peptides

Cross-links involving lysines and glutamines are responsible for a variety
of protein fusions

Naturally Occurring Cross-Links and Cyclic Peptides
(SUMMARY)
1. “Protein fusion problem”
2. De Novo sequencing helps, but more difficult for longer peptides
3. Specialized database search algorithms are required
4. Use of multiple fragmentation techniques and MS3 is a routine in cross-linking analysis
5. -S-S- specific: reversibility and affinity for electrons can be exploited

Protein-Protein contacts: General Approach using CXMS
Majority of current reports in the literature used these platforms:•MALDI-TOF•LTQ•LTQ-Orbitrap-Velos - ETD
Identify cross-linked peptidesMALDI-proTOF Peptide Mass Fingerprint use 2 different proteases, e.g. Proteinase K, and Trypsin
Compare cross-linked monomers to non-cross-linked controlsCompare cross-linked monomers to cross-linked dimers (heterodimers)(any new high-mass, high intensity peaks appearing in cross-linked dimers are
candidate inter-cross-linked peptides)Identify candidate peptide sequencesGenerate all possible peptide pairs (theoretical cross-links)Match found masses to the list
Identify amino acid residues modified with cross-linking reagentIn addition to data-dependent acquisition scheme on LTQ-Orbitrap Velos, use
inclusion list (masses from the comparisons of MALDI-TOF PMF data)Also, use ETD fragmentation feature, in the event of incomplete fragmentation

Formaldehyde: Universal Cross-linking Agent
Briefly,

Formaldehyde: First Step

Formaldehyde: Second Step

Formaldehyde: Pros and Cons
Pros: universality. Cross-links to nucleic acids as wellReversible cross-link (heat above 80 degrees C)
Cons: diverse set of products difficult to find site of cross-linking/interpret mass-spec data
Despite universality, dependences of cross-linking rates on primary/secondary protein structure – variety of conditions need to be screened on case-by-case basis
Cross-linking itself may affect the interaction of interest

24
Cross-Linking Reagents: Choices
Other options:
1) X-linkers with affinity handles
2) MS/MS reporter ions3) Reversible cross-links**
***
*(~2-3 Å)
Complicated chemistry
Too long for precise interface mapping
**

Homobifunctional Cross-Linkers
Trick Question: why TSAT and TMEA spacer arm lengths are so different, while structures are nearly indentical?
piercenet.com

Heterobifunctional Cross-Linkerspiercenet.com

Homework:
1. What is the name of this molecule?2. What is its MW?3. What is its Spacer Arm length?4. What does it cross-link to what?
(circle the reactive moieties and indicate what do they react with)

Photoreactive Amino Acids
piercenet.com

Cross-linked Peptides Enrichment and Detection
O18 method – counts number of C-termini
Affinity handles
Characteristic ions
Sctrong-cation exchange
High-charge driven acquisition

8.017 Da shift (ideally)retention time the same (ideally)
non-ideal situations:1) incomplete labeling2) additional labeling not at C-term (during
deamidation of N and Q, exchange on Y)
Digest(trypsinor other protease)
4.0085 Da shift per C-terminus (2 oxygen atoms exchanged
XC16O2H
C16O2HX
C18O2H
C18O2H
18O –Labeling Method
1) Dry2) Incubate with trypsin in
H218O
3) Inactivate trypsin4) Mix light and heavy 1:1
Un-cross-linked control. ~60 kDa and ~15 kDA protein pair

31
4.0085 Da
FKDLGEEHFK
MSMS data;IT analyzer, CID fragmented
Mass-spec data: regular peptide
2 x 18O atoms
MS/MS (16O)

Mass-spec data: X-linked peptide
8.017 Da 4 x 18O atoms
High res. MS/MS

Multifunctional Cross-LinkersBiotin-Aspartate-Rink-Glycine
MS labile cross-linking reagent,BDRG (biotin-aspartate-Rink-glycine)

Multifunctional Cross-LinkersBiotin-Aspartate-Rink-Glycine
Luo et al 2012 MCP

Multifunctional Cross-LinkersBiotin-Aspartate-Rink-Glycine
Luo et al 2012 MCP

Multifunctional Cross-LinkersBiotin-Aspartate-Rink-Glycine
Luo et al 2012 MCP

Multifunctional Cross-LinkersBiotin-Aspartate-Rink-Glycine
Luo et al 2012 MCP

Mono-Linked Species Detection With MS Labile Cross-Linkers
Luo et al 2012 MCP

Multi-Subunit Complex Architecture
Luo et al 2012 MCP

Cross-linking as a Complimentary Structure-Determination Technique
Luo et al 2012 MCP
Lys-1102 clashes with the noncoding DNA base at the 3 position.helix 37 is parallel to the dissociated noncoding DNA backbone. helix 37 and the following loop (residues 1107–1114 in Rpb1) form a palm-like structure holding the noncoding DNA, with Lys-1102 and Asn-1110 functioning as two fingers guiding the noncoding DNALys-1102 also functions to block the noncoding DNA extending toward the bridge helix, which binds to the template DNA and active center. Luo et al suggest that Lys-507 in fork loop 2, Lys-1102 in the helix 37, andAsn-1110 define an exit path for the noncoding DNA.

Large-Scale Cross-Linking Studies: Examples are Few

Large-Scale Cross-Linking Studies: Examples are Few
Rinner et al study: used DSS, labeled with deuterium (d0/d12 pair); high-charge driven acquisition; strong-cation exchange; novel algorithm - xQuest
E. Coli lysate

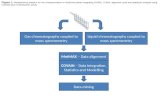
Cross-linking with regular search engine: x Comb Approach
Panchaud et al 2009

Summary
Cross-linking challenges:
1) Physicochemical properties are different for cross-linked peptides2) Incomplete reactions/complicated chemistry3) Difficult to do on the whole proteome scale
Cross-linking strategies:
•There are many choices. Use several cross-linkers suited for a task at hand.•Affinity handles•MS-labile bonds and reversible cross-links•High-charge driven acquisition•High-resolution MS/MS and multiple fragmentation modes•SCX and C4 enrichment•In vivo cross-linking using photoactive amino acids•Count C-termini with O18•Use isotopically-coded cross-linkers
State of the Field: Cross-linking is notoriously difficult and case-specific; despite major advances in instrumentation, there are only few examples of large-scale studies