Chapter 8 - Aldehyde & Ketone
-
Upload
devendran-mahendhren -
Category
Documents
-
view
95 -
download
3
description
Transcript of Chapter 8 - Aldehyde & Ketone
-
Nik Nur Shamiha Nik Dzulkefli
Pharmaceutical Chemistry Unit
SPH
-
Why this chapter?
Aldehydes and ketones are
intermediates in the synthesis of many
pharmaceutical agents, in almost all
biological pathways, and in numerous
industrial processes.
So, an understanding of their properties
and reactions is essential!
-
Chapter Outline Carbonyl Compound
-
Aldehyde & Ketone in Daily Life
Aldehyde and ketones are responsible for many flavors and
odors that you will readily recognize :
-
INTRODUCTION
Aldehyde contain the carbonyl group a group in
which a carbon atom has a double bond to oxygen :
Carbonyl group
Oxygen Carbonyl
Carbon Carbonyl
The carbonyl group in aldehyde is bonded to at
least one hydrogen atom.
Using R, we can designate the general formula as:
C
O
R Hor RCHO ( R = alkyl or aryl or H)
C
O
R H
-
INTRODUCTION
Ketone: the carbon atom in the carbonyl group is
bonded to two hydrocarbon groups :
C
O
R'R
R, R' = substituents
= alkyl or aryl
-
NOMENCLATURE
o IUPAC
o Common name
-
IUPAC Names of Aldehydes
-
Common Names of Aldehydes
In the common system, aldehydes are named with ending aldehyde
C
O
HH C
O
HCH3C
O
H
Formaldehyde Acetaldehyde Benzaldehyde
-
a. IUPAC Names of Ketones
-
b. Common Names of Ketones
-
PHYSICAL PROPERTIES
a. Boiling Point
b. Solubility
-
Physical Properties PROPERTY OBSERVATION
Boiling Point
Solubility
RCHO having 5 Cs are H2O soluble because they can form hydrogen bond with H2O.
RCHO having > 5 Cs are slightly soluble in H2O.
+
-
H O
H + Hydrogen bond with water.
-
Physical Properties
Bp, Mp & Solubility of Aldehydes :
-
Physical Properties
Bp, Mp & Solubility of Ketones :
-
Physical Properties
Boiling Points and Solubility of Aldehydes and Ketones
The carbonyl group is strongly polar but does not produce hydrogen bonding (It has no polar hydrogens). As a result, due to the dipole-dipole interaction, the boiling points of aldehydes and ketones are higher than the nonpolar hydrocarbons and the alkyl halides but lower than those of alcohols.
Formaldehyde is gas at room temperature (b.p. = -21 C) but heavier aldehydes are liquids. Acetone, the simplest ketone, is liquid at room temperature (b.p. = 56 C).
Lower molecular weight aldehydes and ketones are water soluble. Acetone, formaldehyde and acetaldehyde are miscible in water.
-
Physical Properties
Hydrogen bonding typically occurs when a hydrogen atom bonded to O, N, or F, is
electrostatically attracted to a lone pair of
electrons on an O, N, or F atom in another
molecule.
Polar hydrogens
-
Preparation
Preparation of Aldehydes: 1. Oxidation of 1 alcohol 2. Reduction of Acyl Chlorides 3. Partial reduction of Ester
Preparation of Ketones: 1. Oxidation of 2 Alcohol 2. Friedel Crafts Acylation
-
Preparation of Aldehydes: 1. Oxidation of 1 alcohol 2. Reduction of Acyl Chlorides 3. Partial reduction of Ester
Preparation
-
A) Oxidation of 1o Alcohols
General formula:
Using PCC as oxidizing agent :
PCC: Pyridinium chlorochromate
-
A) Oxidation of 1o Alcohols
Using strong oxidizing agent:
CH3CH2OH CH3 C OH
O
Ethanol Ethanoic Acid
H2CrO
4
acetone35oC
CH3CH2OH CH3 C OH
O
Ethanol Ethanoic Acid
KMnO4/ H+
-
B) Reduction of Acyl Chlorides
R C
O
Cl
acid chloride lithium aluminium tri(t-butoxy)hydride
Li+ -
AlH(O-t-Bu)3R C
O
H
aldehyde
CH3CHCH2C
CH3 O
Cllithium aluminium tri(t-butoxy)hydride
Li+ -
AlH(O-t-Bu)3CH3CHCH2C
CH3 O
H
Example:
* Lithium aluminium tri(t-butoxy)hydride is a milder reducing agent that reacts faster with acid chlorides than with aldehydes.
-
CO
Cl
LiAlH(O-t-Bu)3
CO
H
LiAlH(O-t-Bu)3
benzoyl chloride benzaldehyde
CH3CHCH2C
O
Cl
CH3
CH3CHCH2C
O
H
CH3
isovaleryl chloride isovaleraldehyde
B) Reduction of Acyl Chlorides
-
C) Partial reduction of Ester
Certain carboxylic acid derivatives can be partially
reduced to yield aldehydes
Partial reduction of an ester by diisobutylaluminium
hydride (DIBAH) is an important lab-scale method of
aldehyde synthesis, and mechanistically related
processes also occur in biological pathways
1. DIBAH, toluene, -78C
2. H3O+ H
Methyl dodecanoate Dodecanal
-
Preparation
Preparation of Ketones: 1. Oxidation of 2 Alcohol 2. Friedel Crafts Acylation
-
A) Oxidation of 2 Alcohol
Examples
Ketones can be made from 2o alcohols by oxidation
* [O] =
-
B) Friedel Crafts Acylation
Aromatic ketones can be made by Friedel-Crafts Acylation
Examples
-
Reaction of Aldehydes: 1. Oxidation 2. Reduction 3. Nucleophilic Addition 4. Reaction with Grignard reagent
Reaction of Ketones: 1. Reduction 2. Nucleophilic Addition 3. Reaction with Grignard reagent
Reaction
-
Reaction of Aldehydes: 1. Oxidation 2. Reduction 3. Nucleophilic Addition
Reaction
-
1) Oxidation of Aldehydes
Aldehydes are easily oxidized to carboxylic acid by: strong oxidizing agent such as potassium permanganate,KMnO4 mild oxidizing agent such as silver oxide, Ag2O in aqueous ammonia (Tollens Test : differentiate between aldehyde & ketone)
General Reaction
R
C
H
O
[o]
R OH
O
Aldehyde Carboxylic Acid
CH3CH2CH2CH2COH
[O] :
KMnO4, OH-
K2Cr2O7/H2SO4 Ag(NH3)2
+OH- (Tollens reagent)
K2Cr2O7
H2SO4
=
O
CH3CH2CH2CH2CH
=
O
Pentanal Pentanoic acid
Examples
-
1) Oxidation of Aldehydes
In the laboratory, Tollens test may be used to distinguish between an aldehyde and ketone. Tollens reagent, a solution of Ag+ (AgNO3) and ammonia, oxidizes aldehyde, but not ketones. The silver ions is reduced
to metallic silver, which forms a layer called a silver mirror on the inside of the container
* Tollens test is used to distinguish aldehydes from ketones. Ketones DO NOT react with Tollenss reagent.
Tollens Test (Silver Mirror Test)
-
2) Reduction of Aldehydes
Hydride ionLithium aluminum
hydride (LAH)
Sodium
borohydride
H
H H
H
H- B- H H- Al- HLi +Na+
H:
Reduction of an aldehyde gives a
primary alcohol .
Aldehydes can be reduced to alcohol
by
H2/Ni or H2/Pd
LiAlH4 NaBH4
(most often used)
-
2) Reduction of Aldehydes
Examples:
CH3 C H
O
CH3 C H
O-
H
CH3 C H
OH
H
H+
ethanal
ethanol
LiAlH4
-
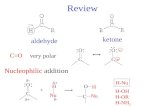
3) Nucleophilic Addition
Nucleophilic addition an addition reaction initiated by attack by an electron-rich reagent (a nucleophile) on a
carbonyl compound or derivative
The carbonyl groups in aldehydes and ketones are polarised because of the difference in the electronegativity of carbon and
oxygen.
The carbon atom carries a partial positive charge while oxygen atom carries a partial negative charge.
Aldehydes and ketones are susceptible to attack both by nucleophiles at the carbonyl carbon atom and by electrophiles
at the oxygen atom.
C O
electrophilic attack nucleophilic attack
- +
-
3) Nucleophilic Addition
Nucleophilic Addition Reaction Of
a. HCN: Cyanohydrin Formation
b. Grignard Reagent : Formation of Alcohol
-
3(a) Nucleophilic addition of hydrogen cyanide
C
O
R H HCN CR R'
OH
CNaldehyde
cyanohydrin
example
C
O
CH3 H HCN CCH3 H
OH
CNethanal
1-hydroxy-1-methylpropanenitrile
* Cyanohydrin may be formed using liquid HCN with a catalytic
amount of sodium cyanide or potassium cyanide.
-
3(b) Reaction With Grignard Reagent
A Grignard reagent (a strong nucleophile resembling a carbanion, R:- attacks the electrophilic carbonyl carbon atom to give an alkoxide intermediate.
Subsequent protonation gives an alcohol.
MgBrCH3CH2C O
H3C
HC O- +MgBr
CH3
H
CH3CH2
C OH
CH3
H
CH3CH2
H3O+
2-butanol
alkoxideethanalethylmagnesium bromide
Grignard reagents react with aldehyde
to give secondary alcohol
-
Reaction of Ketones: 1. Reduction 2. Nucleophilic Addition 3. Reaction with Grignard reagent
Reaction
-
Ketones can be reduced to alcohols using:
a) lithium aluminium hydride (LiAlH4)
b) sodium borohydride (NaBH4)
c) catalytic hydrogenation
H+ = diluted acid such as H2SO4
R C R'
O-
H
R C R'
OH
H
H+
2o alcohol
R C R'
O
LiAlH4 or NaBH4 or H2, Niketone
CH3 C CH3
O-
H
H+
2-propanol
CH3 C CH3
O
H2/Ni
propanone
CH3 C CH3
OH
H
Example:
1. Reduction to Secondary Alcohols
-
CO
R R' HCN
C
O
CH3 CH3 HCN
CR R'
OH
CN
CCH3 CH3
OH
CN
ketone
cyanohydrin
example
propanone
2-hydroxy-2-methylpropanenitrile
* Cyanohydrin may be formed using liquid HCN with a
catalytic amount of sodium cyanide or potassium cyanide.
2. Nucleophilic addition of hydrogen cyanide
-
CO
R R' HCN
C
O
CH3 CH2CH3 HCN
CR CN
OH
R'
CCH3 CN
OH
CH2CH3
ketone
cyanohydrin
example
propan-2-one
H2O/H+
CR COOH
OH
R
a-hydroxyacid
NH4+
H2O/H+
CCH3 COOH
OH
CH2CH3
NH4+
'
Cyanohydrin can be hydrolysed to give -hydroxyacids.
The nitrile (-CN) group is converted to the COOH group by reflux the cyanohydrin with dilute sulphuric acid (H2O/H
+) or
concentrated HCl.
2 Nucleophilic addition of hydrogen cyanide
-
A Grignard reagent (a strong nucleophile resembling a
carbanion, R:- attacks the electrophilic carbonyl carbon atom to
give an alkoxide intermediate.
Subsequent protonation gives an alcohol.
MgBrCH3CH2C O
H3C
H3CC O- +MgBr
CH3
CH3
CH3CH2
C OH
CH3
CH3
CH3CH2
H3O+
2-methyl-2-butanol
alkoxideacetoneethylmagnesium bromide
3. Reaction with Grignard Reagent
Grignard reagents react with ketones
to give tertiary alcohol
-
Principles uses of Aldehyde & Ketone
-
Aldehydes and ketones are used as solvents, starting materials and reagents for the synthesis of other products.
Formaldehyde is well recognized as formalin solution used to defend biological specimens and to prepare, urea-formaldehyde glues and other polymeric products.
Aldehyde is used primarily as an initial material in the manufacture of acetic acid, ethyl acetic, vinyl acetate, polymers and drugs.
Benzaldehyde is used in perfumery with in dye industries. Acetone with ethyl methyl ketones are general industrial solvents.
Various aldehydes and ketones exemplar, butyraldehyde, vanillin, acetophenone, camphor, etc. are well recognized in support of their odours and flavours.
Principles uses of Aldehyde & Ketone
-
Aldehyde & Ketone in Daily Life
Aldehyde and ketones are responsible for many flavors and odors that you will readily recognize :
-
Tests to Distinguish Aldehydes and Ketones, and Aliphatic
Aldehydes and Aromatic Aldehydes
TESTS ALDEHYDES KETONES
Tollens Test / silver mirror test Reagent and condition:
- ammoniacal silver nitrate
solution ([Ag(NH3)2]+)
Observation:
Formation of silver mirror
Observation:
Silver mirror did not formed
* Ketones do not react with
Tollens reagent
Fehlings test / Benedicts test Reagent and condition:
-Solution of Cu2+ (aq) ions in an
alkaline solution of sodium
potassium tartate.
*Can be used to distinguish
between:
i) Aldehydes and ketones
ii) Aliphatic aldehydes and
benzaldehyde
Observation;
Blue colour of the Fehlings solution dissappears and
brick-red precipitate is
obtained
* Except benzaldehyde
Observation:
Blue colour remains.
* Ketones do not react with
Fehlings/Benedicts reagent
Schiffs test Reagent and condition:
- Schiffs reagent
Observation:
Formation of magenta-pink
colour (simple aldehydes)
* Except benzaldehyde and
a few aromatic aldehydes)
Observation:
Ketones (except propanone)
do not react with Schiffs reagent.
-
End of Chapter 8