(Chapter 7). - Overview - Classification and Structure of Carbohydrates : A) Isomers and epimers B)...
-
Upload
shawn-benson -
Category
Documents
-
view
227 -
download
0
Transcript of (Chapter 7). - Overview - Classification and Structure of Carbohydrates : A) Isomers and epimers B)...

- Overview
- Classification and Structure of Carbohydrates :
A) Isomers and epimers
B) Enantiomers
1st Lecture: Pages : 83 - 84

:OVERVIEW
Carbohydrates are the most abundant organic molecules in nature.
They have a wide range of functions including :
- providing a significant fraction of the energy in the diet of most organisms
- acting as a storage form of energy in the body
- serving as cell membrane components mediate some forms of intercellular communication.

Carbohydrates serve as a structural component of many organisms:
- the cell walls of bacteria - the exoskeleton of many insects - the fibrous cellulose of plants.
The empiric formula for many of the simpler carbohydrates is (CH2O)n.
hence the name “hydrate of carbon”.



CLASSIFICATION AND STRUCTURE OF CARBOHYDRATES:
Monosaccharides :- Monosaccharides (simple sugars) can be
classified according to the number of carbon atoms they contain as trioses, tetroses, pentoses, hexoses or heptoses.
. - Examples of some monosaccharides
commonly found in humans are listed in (Figure7.1)





CH
O
reducinggroup
OH
OH
OH
OH
H
H
H
H
C
C
C
C
CH2OH
1
2
3
4
5
6
carbon
D-Glucose(open chain)
Structure of glucose in its linear form

OH
OH
OH
OH
H
HH
H
H CC
CC
C
O
CH2OH6
1
O
CH2OH
OH
OH
OH
OH
6
1
- anomer of glucose
O
CH2 OHOH
OH
OH
OH
6
1
- anomer of glucose
Glucose in its ring forms









Aldoses and Ketoses:
• Carbohydrates with an aldehyde group as their most oxidized functional group are called aldoses.
• Carbohydrates with a keto group as their most oxidized functional group are called ketoses (Figure7.2).



Monosaccharides are classified to Aldose or Ketose depending upon they have an aldehyde or ketone group.
Aldose Ketose Tetrose (C4H8O4) Erythrose ErythulosePentose (C5H10O5) Ribose RibuloseHexose (C6H12O6) Glucose Fructose
• Carbohydrates that have a free carbonyl group have the suffix “-ose “
[ Note: Ketoses (with some exceptions e.g. fructose) have an additional two letters in their suffix: “-ulose” for example, xylulose].

• Monosaccharides : - Carbohydrates that can not be hydrolyzed into
simpler CHO. - Can be linked by glycosidic bonds to create
larger structures (Figure7.3).
• Disaccharides: contain two monosaccharide units e.g.:
- Lactose ( galactose + glucose ). - Maltose ( glucose + glucose ). - Sucrose ( glucose + fructose ).



O
CH2OH
O
O CH 2 OH
SUCROSE (glucose-fructose)
-1,2-bond
OH
CH2OH
O O
O
LACTOSE (galactose-glucose)
-1,4-bond
OH
OH CH2OH CH2OH
O O
O
MALTOSE (glucose-glucose)
OH OH
CH2OH CH2OH
-1,4-bond
O O
O OH OH
CH2OH CH2OH
-1,1-bond
TREHALOSE (glucose-glucose)
Structures of the most common disaccharides.
1 2
1 41 1
1 4





• Oligosaccharides : Contain 3 to 12 monosaccharide units
(Maltotriose , Glycoproteins).
• Polysaccharides : Contain more than 12 monosaccharide units,
and can be hundreds of sugar units in length: - Starch : Amylose, Amylopectin,Dextrins
- Cellulose - Glycogen “ homopolysaccharides” - Glycosaminglycan “ heteropolysaccharides”

POLYSACCHARIDES
Cellulose: - linear chain with 1,4 glycosidic bonds
Amylose: - Plant CHO storage - Linear chain with 1,4 glycosidic bonds
Amylopectin: - Amylose + branches with 1,6 glycosidic bonds
Glycogen : - Animal CHO storage - Similar to amylopectin except more branches
Oligosaccharides: - 4-10 (average 8) glucose units; - -limit dextrin = branched (1 or 2 1,6 bonds)


STRUCTURE OF MONOSACCHARIDES:
A) Isomers and polymers:
Isomers : Compounds having the same chemical formula but having different structures e.g. fructose, glucose, mannose, and galactose are all isomers of each other, [ C6H12O6 ].
Epimers : 2 monosaccharides differ in configuration around only one specific carbon atom (with the exception of the carbonyl carbon see “anomers”) [they are also isomers to each other] Example : glucose and galactose are C- 4 epimers , differ only in the position of the - OH group at C- 4]

The carbons in sugars are numbered beginning at the end that contains the carbonyl carbon - that is, the aldehyde or keto group (Fig7. 4).
Glucose and galactose are C-4 epimers.
Glucose and mannose are C-2 epimers.
Galactose and mannose are NOT epimers, they differ in the position of - OH groups at two carbons (2 and 4) and are therefore defined only as isomers.


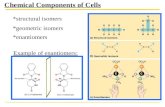
B) Enantiomers:• A special type of isomerism is found in the pairs of
structures that are mirror images of each other.
• The mirror images called enantiomers, and the two members of the pair are designated as a D- and an L-sugar (Figure 7.5).
• The orientation of the - H & - OH groups around the Carbon atom adjacent to the terminal primary alcohol carbon ( C 5 in glucose) determines whether the sugar is D or L sugar.
• D sugar ( -OH group on C5 is on the Right )
• L sugar ( -OH group on C5 is on the Left )
• The majority of the sugars in mammals are D-sugars.

Enantiomers

Thank You

- Classification and Structure of Carbohydrates :
C) Cyclization of Monosaccharides
D) Complex Carbohydrates
- Starch
- Glycogen
- Cellulose
2nd Lecture: Page : 84-85 & 123-124

C) Cyclization of monosaccharides:• < 1% of the monosaccharides with five or more carbons
exists in the open-chain (acyclic form).
• They are predominantly found in a ring form : [ aldehyde (or ketone) group + an alcohol group on the same sugar] hemiacetal or hemiketal ring.
- Aldose hemiacetal
- Ketose hemiketal
• If the resulting ring has six members ( 5 C + 1 O) pyranose ring.
• If the resulting ring has five members ( 4 C + 1 O) furanose ring.

Ring with 5 C + 1 O
Ring with 4 C + 1 O

1) Anomeric carbon:
• Formation of a ring an anomeric carbon at C 1 of an aldose or at C 2 of a ketose.
• These structures or configurations of the sugar, e.g.. -D- glucose and -D-glucose (Figure.7.6).
• These 2 sugars are both glucose, but they are anomers of each other.
• Glycogen is synthesized from - D - glucopyranose, Cellulose is synthesized from - D - glucopyranose.

• The cyclic and anomers of a sugar in solution are in equilibrium with each other and can be interconverted, a process called mutarotation. (Figure7.6)
• Crystalline glucose is an - D - glucopyranose.
• Glucose in solution keeps its cyclic structure but isomerism occurs at the anomeric carbon atom (C1)
- - glucopyranose (38 %)
- - glucopyranose (62 %)
- and anomers of glucofuranose ( < 0.3 % )

Cyclization of monosaccharides

2) Reducing sugars: • If the oxygen on the anomeric carbon (the carbonyl group)
of a sugar is not attached to any other structure reducing sugar
• A reducing sugar can react with chemical reagents (e.g.. Benedict’s solution) and reduce the reactive component. The anomeric carbon becoming oxidized
[ Note: Only the state of the oxygen on the anomeric carbon determines if the sugar is reducing or nonreducing - the other -OH groups on the molecule are not involved ].

D. Complex carbohydrates:• Carbohydrates attached to non-carbohydrate structures by
glycosidic bonds “Complex carbohydrates”.
• The non carbohydrate portion named [ aglycone ] e.g.:
- Purines and pyrimidines (Nucleic acids).
- Aromatic rings (Steroids and bilirubin).
- Proteins (Glycoproteins and glycosaminoglycans)
- Lipids (Glycolipids)
• The entire molecule by the generic name called [ glycoside ]

• The C 1 of aldose, or the C 2 of Ketose participating in the glycosidic link, is called [ a glycosyl residue ].
• if the anomeric carbon of glucose participates in such a bond, that sugar is called a glucosyl residue. (Figure. 7.3).
• If the identity of the attached sugar is known glucoside or galactoside. [ If anomeric carbon of a sugar is in a glycosidic linkage no longer a reducing sugar ].


Polysaccharides:
1. Macromolecules containing hundreds or thousands of monosaccharides linked together.
2. Storage polysaccharides : Starch – made entirely of glucose.
3. Structural polysaccharides : include cellulose and chitin:
- Cellulose most abundant organic compound on earth.
- Cellulose is a polymer of glucose (like starch) but linkage between glucose monomers differ :
- in Starch , alpha () linkage form
- in Cellulose , beta () linkage form

• The major polysaccharides are of animal origin (Glycogen) and plant origin ( Starch) and (Cellulose).
• Starch composed of amylose and amylopectin and contains ( 1 4) glycosidic bonds .
• Cellulose is the most abundant polysaccharide of plant origin in nature composed of hundreds of glucose residues contains -D- glucopyranose linked by ( 1 4) glycosidic bonds.



Starch and cellulose have different shapes and therefore, different properties:
• In cellulose , many parallel glucose strands held together by H bonding.
• These parallel strands form microfibrils.
• Microfibrils intertwine to form a cellulose fibril.
• Microfibrils are then super coiled to form cables of cellulose

• Cellulose not digested by human beings, but aids in production of mucous along the walls of the intestinal tract facilitates passage of food.
• Few organisms (bacteria , molds) produce enzymes which digest cellulose

1. O- and N-glycosides:
• If the group on the non-carbohydrate molecule (aglycone) to which the sugar is attached is an -OH group O-glycoside.
• If the group is an -N H2 N-glycoside (Figure7.7).
[Note: All sugar-sugar glycosidic bonds are O-type linkages].
• Typical aglycones :
- purines and pyrimidines ( in nucleic acids) - aromatic rings (in steroids and bilirubin) - proteins (in glycoproteins and glycosaminoglycans) - lipids ( in glycolipids).

Complex CHO : Glycosides



2) Naming glycosidic bonds:
• Glycosidic bonds between sugars are named according to :
- the numbers of the connected carbons - the position of the anomeric -OH group of the sugar involved in the bond.
• If anomeric -OH group is in the - configuration, the linkage is an - bond.
• If it is in the - configuration, the linkage is a -bond.




Structure and function of glycogen: • Glycogen is a branched – chain homopolysaccharide made
exclusively from -D-Glucose.
• The primary glycosidic bond is (1 4) linkage.
• After 8-10 glycosidic residues, there is a branch containing an (1 6) linkage.
• A single molecule of glycogen have a molecular mass of up to 108 daltons.
• Glycogen molecules exist in discrete cytoplasmic granules that contain most of the enzymes need for glycogen synthesis and degradation (Figure 11.3).




• The main stores of glycogen are skeletal muscles and liver
• The function of muscle glycogen is to serve as a fuel reserve for synthesis of ATP during muscle contraction
• The function of liver glycogen is to maintain the blood glucose level during early stages of fasting (Figure 11.2)
• 4oo g of glycogen makes up to 1 – 2 % of weight of resting muscle.
• 1oo g of glycogen makes up to 10 % of fresh weight of well–fed adult liver.


• In some glycogen-storage diseases (e.g. Pompe disease, VonGierke's disease & McArdle syndrome), the amount of glycogen in the liver and/or muscle can be significantly higher.
• Liver glycogen stores during the well – fed state and are depleted during fasting state
• Muscle glycogen is not affected by short period of fasting and is only moderately in prolonged fasting.
• Muscle glycogen is synthesized to replenish muscle stores after its depletion following strenuous exercise.


• Lactose, for example, is synthesized by forming a glycosidic bond between C 1 of a -galactose and C 4 of glucose.
• The linkage is, therefore, a (1 4) glycosidic bond. (see Figure 7.3).
• Cellulose contains ( 1 4) glycosidic bonds and is the most abundant polysaccharide of plant origin in nature composed of hundreds of glucose residues
[Note: Because the anomeric end of the glucose residue is not involved in the glycosidic linkage it (and Therefore, lactose) remains a reducing sugar].

Cellulose and Starch have different shapes and therefore, different properties:
• In cellulose , many parallel glucose strands held together by H bonding.
• These parallel strands form microfibrils.
• Microfibrils intertwine to form a cellulose fibril.
• Microfibrils are then super coiled to form cables of cellulose

• Cellulose not digested by human beings, but aids in production of mucous along the walls of the intestinal tract facilitates passage of food.
• Few organisms ( bacteria , molds) produce enzymes which digest cellulose


Thank you