Chapter 14faperta.ugm.ac.id/download/bahan_kuliah/tutik_dw/Kimia_Organik/CH... · Organic Chemistry...
Transcript of Chapter 14faperta.ugm.ac.id/download/bahan_kuliah/tutik_dw/Kimia_Organik/CH... · Organic Chemistry...

Organic Chemistry OnLine ©2000
Chapter 14
Electrophilic Aromatic Substitution-II
© 2000, Paul R. YoungUniversity of Illinois at Chicago
All Rights Reserved

Organic Chemistry OnLine ©2000
Electrophilic Aromatic Substitution
Acylation
Alkylation
Sulfonation
Nitration
Halogenation
C
O
R
R
SO3H
NO2
X X+
NO2+ HSO3
+ R+
RC O+

Organic Chemistry OnLine ©2000
Br2/FeBr3
Br
Fe
Br
Br
Br
Br Brδ δ
FeBr3 functions as a Lewis Acid, binding Br2 andcreating a source of the bromonium cation, Br+

Organic Chemistry OnLine ©2000
Br
Br
H
Br
H
H
Br
Br loss of H+
...the driving force for loss of H+ is therestoration of the aromatic system.

Organic Chemistry OnLine ©2000
Br
H
Br
H
H
Br
An electrostatic potential map showing chargedistribution in the cyclohexadienyl cation intermediate.
Electron donatingsubstituents at these
positions will stabilizethe cationic
intermediate.

Organic Chemistry OnLine ©2000
Cl2/FeCl3
ClCl
H
II
HI2
CuCl2
I
...CuCl2/I2 serves to generate I+

Organic Chemistry OnLine ©2000
NO2
NO2+
NO2
H
H2SO4 + HNO3 H2O + NO2+
Nitration

Organic Chemistry OnLine ©2000
SO3H
H
H2SO4 + SO3 SO3H+
+ HSO4-
SO3H+
SO3H
Sulfonation

Organic Chemistry OnLine ©2000
Friedel-Crafts Alkylation
CH3
CH3
HCH3
Cl
AlCl3
δ
δ
δδ
CH3 Cl AlCl3CH3Cl + AlCl3

Organic Chemistry OnLine ©2000
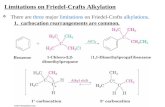
Limitations of the Friedel-Crafts Alkylation
1. Reaction limited to alkyl halides; aryl or vinyl halides donot react.
2. Reaction does not occur on rings containing strongelectron withdrawing substituents.
3. Multiple substitutions often occur.
4. Carbocation rearrangements can occur, particularly with1˚ alkyl halides.
NO2 CN SO3H CHO COR
COOH COOR NR3+

Organic Chemistry OnLine ©2000
O
CH3H
O
CH3
CH3
O
Friedel-Crafts Acylation
Cl AlCl3
CH3
O δ δ
ClH3C
O
+ AlCl3

Organic Chemistry OnLine ©2000
Friedel-Crafts Acylation
1. Multiple substitutions do not occur.
2. Carbocation rearrangements do not occur.
3. Reaction does not occur on rings containing strongelectron withdrawing substituents.
4. Acid anhydrides can also be used.
O
CH3
...ring is deactived after mono-substitution;multiple substitutions do not occur.
H3C O CH3
O O

Organic Chemistry OnLine ©2000
1. Predict the products of the following reactions.
AlCl3
AlCl3
Cl
Cl
O
HNO3/H2SO4

Organic Chemistry OnLine ©2000
1. Predict the products of the following reactions.
AlCl3
AlCl3
Cl
Cl
O
HNO3/H2SO4
O
CH3

Organic Chemistry OnLine ©2000
1. Predict the products of the following reactions.
AlCl3
AlCl3
Cl
Cl
O
HNO3/H2SO4
O
CH3
NO2

Organic Chemistry OnLine ©2000
1. Predict the products of the following reactions.
AlCl3
AlCl3
Cl
Cl
O
HNO3/H2SO4
O
CH3
NO2

Organic Chemistry OnLine ©2000
1.(con't) Predict the products of the following reactions.
AlCl3
O
O O
FeCl3
Cl2
SO3/H2SO4

Organic Chemistry OnLine ©2000
1.(con't) Predict the products of the following reactions.
AlCl3
O
O O
FeCl3
Cl2
SO3/H2SO4
Cl

Organic Chemistry OnLine ©2000
1.(con't) Predict the products of the following reactions.
AlCl3
O
O O
FeCl3
Cl2
SO3/H2SO4
Cl
SO3H

Organic Chemistry OnLine ©2000
1.(con't) Predict the products of the following reactions.
AlCl3
O
O O
FeCl3
Cl2
SO3/H2SO4
Cl
SO3H
O
CH2CH3

Organic Chemistry OnLine ©2000
H2/Pd25˚ C, 1 atm
Catalytic Reduction of AromaticCompounds

Organic Chemistry OnLine ©2000
H2/Pd25˚ C, 1 atm

Organic Chemistry OnLine ©2000
H2/Pd25˚ C, 1 atm
H2, Pt/C 25˚ C2000 psi

Organic Chemistry OnLine ©2000
H2/Pd25˚ C, 1 atm
H2, Rh/C25˚ C; 1 atm
H2, Pt/C 25˚ C2000 psi

Organic Chemistry OnLine ©2000
The Reduction of Aryl Nitro Groups and ArylKetones
O
H2/Pd25˚ C, 1 atm
NO2
H2/Pd25˚ C, 1 atm
NH2
...or...SnCl2/H3O
+

Organic Chemistry OnLine ©2000
+
O
Zn(Hg)H3O
+
Aryl ketones can also be reduced to thehydrocarbon using the Clemmensen Reduction.

Organic Chemistry OnLine ©2000
Suggest a synthesis of propylbenzene, beginning with benzene.

Organic Chemistry OnLine ©2000
AlCl3CH3CH2CH2Cl

Organic Chemistry OnLine ©2000
AlCl3CH3CH2CH2Cl

Organic Chemistry OnLine ©2000
Cl AlCl3 AlCl3CH3CH2CH2Cl

Organic Chemistry OnLine ©2000
1,2-hydride shift
Cl AlCl3
Cl AlCl3 AlCl3CH3CH2CH2Cl
H
H

Organic Chemistry OnLine ©2000
1,2-hydride shift
Cl AlCl3
Cl AlCl3 AlCl3CH3CH2CH2Cl
H
H

Organic Chemistry OnLine ©2000
Suggest a synthesis of propylbenzene, beginning with benzene.

Organic Chemistry OnLine ©2000
O
CH3CH2 C
O
Cl
AlCl3

Organic Chemistry OnLine ©2000
AlCl3
H2/Pd25˚ C, 1 atm
CH3CH2 C
O
Cl
O

Organic Chemistry OnLine ©2000
Reactions of Arylamines
H2/Pd25˚ C, 1 atm
NO2NH2
...or...SnCl2/H3O
+

Organic Chemistry OnLine ©2000
Reactions of Arylamines
H2/Pd25˚ C, 1 atm
NO2NH2
...or...SnCl2/H3O
+
NH2 N
N
HNO2H2SO4
A diazonium salt.

Organic Chemistry OnLine ©2000
H3PO2, H 2O
HCl, CuCl
HBr, CuBr
KI
N2+

Organic Chemistry OnLine ©2000
H3PO2, H 2O
HCl, CuCl
HBr, CuBr
KI
N2+

Organic Chemistry OnLine ©2000
H3PO2, H 2O
HCl, CuCl
HBr, CuBr
KI
N2+
Cl
Sandmeyer Reaction

Organic Chemistry OnLine ©2000
H3PO2, H 2O
HCl, CuCl
HBr, CuBr
KI
N2+
Cl
Br

Organic Chemistry OnLine ©2000
H3PO2, H 2O
HCl, CuCl
HBr, CuBr
KI
N2+
Cl
Br
I

Organic Chemistry OnLine ©2000
KCN, CuCN
H+/H2O
OH
N
CH3
CH3
N2+

Organic Chemistry OnLine ©2000
KCN, CuCN
H+/H2O
OH
N
CH3
CH3
N2+
CN

Organic Chemistry OnLine ©2000
KCN, CuCN
H+/H2O
OH
N
CH3
CH3
N2+
CN
OH

Organic Chemistry OnLine ©2000
KCN, CuCN
H+/H2O
OH
N
CH3
CH3
N2+
CN
OH
NN
OH

Organic Chemistry OnLine ©2000
OH
The oxygen atom in phenol is electron-releasing by resonance,activating the ring to electrophilic aromatic substitutionortho- and para- to the oxygen.

Organic Chemistry OnLine ©2000
OH
The oxygen atom in phenol is electron-releasing by resonance,activating the ring to electrophilic aromatic substitutionortho- and para- to the oxygen.
OH

Organic Chemistry OnLine ©2000
OH
The oxygen atom in phenol is electron-releasing by resonance,activating the ring to electrophilic aromatic substitutionortho- and para- to the oxygen.
OH
OH

Organic Chemistry OnLine ©2000
OH OH
OHOH
The oxygen atom in phenol is electron-releasing by resonance,activating the ring to electrophilic aromatic substitutionortho- and para- to the oxygen.

Organic Chemistry OnLine ©2000
OHδ
δ δ
Because of the increased electron density, an electrophile willattack the phenol ring preferentially at these positions.

Organic Chemistry OnLine ©2000
OHδ
δ δ
Because of the increased electron density, an electrophile willattack the phenol ring preferentially at these positions.
NN
A diazonium salt acting asan electrophile.

Organic Chemistry OnLine ©2000
NN
OH
OHδ
δ δ
NN
H+
OH
NN
HH
Because of the increased electron density, an electrophile willattack the phenol ring preferentially at these positions.

Organic Chemistry OnLine ©2000
NN
OH
OHδ
δ δ
NN
H+
OH
NN
HH
Because of the increased electron density, an electrophile willattack the phenol ring preferentially at these positions.
A diazonium“coupling” product.

Organic Chemistry OnLine ©2000
KCN, CuCN
H+/H2O
OH
N
CH3
CH3
N2+
CN
OH
NN
OH

Organic Chemistry OnLine ©2000
KCN, CuCN
H+/H2O
OH
N
CH3
CH3
N2+
CN
OH
NN
OH
NN
N
CH3
CH3

Organic Chemistry OnLine ©2000
NH2 NH2
Br
Br
Br
Br2
OH
OH
Br
Br
Br
Br2
Arylamines and Phenols are so highly activated towardselectrophilic aromatic substitution that they react withbromine in the absence of a catalyst to give tribromo adducts.

Organic Chemistry OnLine ©2000
Reactivity of Substituted Benzenes
1. Substituents effect the reactivity of aromatic rings.
2. Substituents also effect the orientation orregiochemistry of the reaction.

Organic Chemistry OnLine ©2000
NO2CN
SO3H
CHO
CORCOOHCOOR
NR3+
NH2
OCH3
OH
N
H
C
O
CH3CH3
F
BrCl
I
H
stronglyactivating
stronglydeactivating

Organic Chemistry OnLine ©2000
Electrostatic potential maps clearly show the effect ofsubstituents on electron density in a substituted benzene ring.
Aniline, stronglyactivating
Benzene Benzonitrile,strongly deactivating

Organic Chemistry OnLine ©2000
Electrostatic potential maps clearly show the effect ofsubstituents on electron density in a substituted benzene ring.
Aniline, stronglyactivating
Benzene Benzonitrile,strongly deactivating

Organic Chemistry OnLine ©2000
Electrostatic potential maps clearly show the effect ofsubstituents on electron density in a substituted benzene ring.
Aniline, stronglyactivating
Benzene Benzonitrile,strongly deactivating

Organic Chemistry OnLine ©2000
Electrostatic potential maps clearly show the effect ofsubstituents on electron density in a substituted benzene ring.
Aniline, stronglyactivating
Benzene Benzonitrile,strongly deactivating

Organic Chemistry OnLine ©2000
δ
OCH3
δ
δ
δ
E
OCH3
OCH3 OCH3 OCH3

Organic Chemistry OnLine ©2000
Br
H
Br
H
H
Br
Electron donatingsubstituents at these
positions will stabilizethe cationic
intermediate.
Recall that positive charge in the cyclohexadienyl cationintermediate is delocalized to the ortho- and para- positions.

Organic Chemistry OnLine ©2000
δ
OCH3
δ
δ
δ
E
Because of this, attack to form the cationic intermediate willoccur predominately ortho- and para- to an electron donatingsubstituent.
Electron donatingsubstituents at these
positions will stabilizethe cationic
intermediate.

Organic Chemistry OnLine ©2000
ortho ¶
directing
NO2CN
SO3H
CHO
CORCOOHCOOR
NR3+
NH2
OCH3
OH
N
H
C
O
CH3CH3
F
BrCl
I
H
stronglyactivating
stronglydeactivating

Organic Chemistry OnLine ©2000
Cl
Cl Cl Cl
δ
Cl
δ
δ
δ
E ...halogens are deactivating inductively,but activate the ring through resonance.

Organic Chemistry OnLine ©2000
CH3
HNO3/H2SO4
CH3
NO2
CH3
NO2
CH3
NO2
63% 3%
34%
...the alkyl group is activating and increases electron densityin the ortho and para positions; thus, nitration generates
predominately ortho and para products.

Organic Chemistry OnLine ©2000
CH3
H
E
CH3
E
H
CH3
E
H
CH3
+
E
a tertiarycarbocation
...alkyl groups activate ortho and para because the sigma-complex intermediates yield more stable tertiary carbocations.

Organic Chemistry OnLine ©2000
CN
HNO3/H2SO4
CN
NO2
CN
NO2
CN
NO2
17% 81%
2%
...the cyano group is deactivating; nitration generatespredominately meta product.

Organic Chemistry OnLine ©2000
δ
δ
δ
δNO2
NOO
NOO
NOO
E
NOO

Organic Chemistry OnLine ©2000
Recall that electron donatingsubstituents ortho- and para-
to the sigma intermediate willstabilize the cationic
intermediate.
Deactivating meta-directing groups...
δ
δ
δ
δNO2

Organic Chemistry OnLine ©2000
When electron withdrawing substituentsare present ortho- or para- to the sigma
intermediate substitution is forced meta-because there is reduced electron density in
the other positions.
Deactivating meta-directing groups...
δ
δ
δ
δNO2

Organic Chemistry OnLine ©2000
NOO
E
NOO
NOO
NOO
NO2
δ
δ
δ
δ
...electrophilic attack occurs at the metapositions because the ortho and para
positions are electron deficient.

Organic Chemistry OnLine ©2000
ortho- ¶-
directing
NO2CN
SO3H
CHO
CORCOOHCOOR
NR3+
NH2
OCH3
OH
N
H
C
O
CH3CH3
F
BrCl
I
H
stronglyactivating
stronglydeactivating
meta-directing

Organic Chemistry OnLine ©2000
CH3
NO2
HNO3/H2SO4
Orientation Effects in Disubstituted Benzenes

Organic Chemistry OnLine ©2000
CH3
NO2
HNO3/H2SO4
CH3
NO2
NO2
CH3
NO2
NO2O2N
methyl directs ortho
nitro directs meta
Trinitrotoluene

Organic Chemistry OnLine ©2000
CH3
NO2
HNO3/H2SO4
CH3
NO2
NO2
CH3
NO2
NO2O2N
methyl directs ortho
nitro directs meta
TriNitroToluene

Organic Chemistry OnLine ©2000
CH3
OCH3
Br2/FeBr3
Orientation Effects in Disubstituted Benzenes

Organic Chemistry OnLine ©2000
CH3
OCH3
Br2/FeBr3
CH3
OCH3
Br
...when substituents direct to different positions, themost powerful group will dominate.

Organic Chemistry OnLine ©2000
CH3
Cl
Cl2/FeCl3
Orientation Effects in Disubstituted Benzenes

Organic Chemistry OnLine ©2000
CH3
Cl
Cl2/FeCl3
CH3
Cl
Cl
CH3
Cl
Cl
CH3
Cl
Cl
+
...product not observed;too hindered

Organic Chemistry OnLine ©2000
Nucleophilic Aromatic Substitution
F
NO2
NO2
HO-
OH
NO2
NO2

Organic Chemistry OnLine ©2000
F
NO2
O2N
HO-
...SN2 attack is not possible;SN1 would generate the unstable aryl cation.
F
NO2
NO2
NO2
NO2

Organic Chemistry OnLine ©2000
F
NO2
NO2
HO-
NO2
NO2
OHF
NO2
NO2
OH
NO2
NO2
OHF
...addition
...elimination
The Addition-Elimination Mechanism

Organic Chemistry OnLine ©2000
N
NO2
OHF O
ON
NO2
OHF O
O
...the carbanion at the ortho- position is stabilizedbyresonance with the oxygens of the nitro group

Organic Chemistry OnLine ©2000
N
NO2
OHF O
ON
NO2
OHF O
O
+ F
NO2
OH
N
O
O
elimination

Organic Chemistry OnLine ©2000
CH3
SO3H
NO2
Br2/FeBr3
I2/CuCl2
Cl2/FeCl3

Organic Chemistry OnLine ©2000
CH3
SO3H
NO2
Br2/FeBr3
I2/CuCl2
Cl2/FeCl3CH3
Cl + ortho isomer

Organic Chemistry OnLine ©2000
CH3
SO3H
NO2
Br2/FeBr3
I2/CuCl2
Cl2/FeCl3CH3
Cl
SO3HBr
+ ortho isomer

Organic Chemistry OnLine ©2000
CH3
SO3H
NO2
Br2/FeBr3
I2/CuCl2
Cl2/FeCl3CH3
Cl
SO3HBr
NO2I
+ ortho isomer

Organic Chemistry OnLine ©2000
CH3
CH(CH3)2
ClSO3/H2SO4
excess HNO3/H2SO4
Cl
O
C
AlCl3

Organic Chemistry OnLine ©2000
CH3
CH(CH3)2
ClSO3/H2SO4
excess HNO3/H2SO4
Cl
O
C
AlCl3
Cl
HO3S+ ortho isomer

Organic Chemistry OnLine ©2000
CH3
CH(CH3)2
ClSO3/H2SO4
excess HNO3/H2SO4
Cl
O
C
AlCl3
CH3
NO2
O2N NO2
Cl
HO3S+ ortho isomer

Organic Chemistry OnLine ©2000
CH3
CH(CH3)2
ClSO3/H2SO4
excess HNO3/H2SO4
Cl
O
C
AlCl3
CH3
NO2
O2N NO2
CH(CH3)2
O
Cl
HO3S+ ortho isomer