ch14-slides0
-
Upload
soyblahblah -
Category
Documents
-
view
214 -
download
0
description
Transcript of ch14-slides0
-
Chapter 14:
ISSUES TO ADDRESS
Polymer StructuresISSUES TO ADDRESS... What are the general structural and chemical
characteristics of polymer molecules?characteristics of polymer molecules? What are some of the common polymeric
materials, and how do they differ chemically? How is the crystalline state in polymers different
from that in metals and ceramics ?
Chapter 14 - 1
-
What is a Polymer?
Poly mermany repeat unit
C C C C C CHHHHHH
C C C C C CHHHHHH HHH H
C C C C C CHH
repeatunit
repeatunit
repeatunit
C C C C C CHHHHHH
Polyethylene (PE)ClCl Cl
C C C C C CHHH
Poly(vinyl chloride) (PVC)HH
Polypropylene (PP)
C C C C C CCH3CH3CH3H
Adapted from Fig. 14.2, Callister & Rethwisch 8e.
Chapter 14 - 2
-
Ancient Polymers Originally natural polymers were used
Wood Rubber Wood Rubber Cotton Wool
L th Silk Leather Silk
Old t k Oldest known uses Rubber balls used by Incas Noah used pitch (a natural polymer)
for the ark
Chapter 14 - 3
-
Polymer CompositionMost polymers are hydrocarbons
i.e., made up of H and Cp Saturated hydrocarbons
Each carbon singly bonded to four other atoms Example:
Ethane, C2H6
C C
HHH
C C
H H H
Chapter 14 - 4
-
Chapter 14 - 5
-
Unsaturated Hydrocarbons Double & triple bonds somewhat unstable
can form new bonds Double bond found in ethylene or ethene - C2H4
H HC C
H
H
H
H
Triple bond found in acetylene or ethyne - C2H22 2
C C HH
Chapter 14 - 6
-
Isomerism Isomerism
two compounds with same chemical formula cantwo compounds with same chemical formula can have quite different structures
for example: C8H18 normal-octane
C C C C C C C CH
H H H H H H H H
H H3C CH2 CH2 CH2 CH2 CH2 CH2 CH3=
2 4 dimethylhexane
H H H H H H H H
H3C CH2 CH3( )6
2,4-dimethylhexane
H3C CH
CH3CH2 CH
CH
CH3
Chapter 14 - 7
CH2CH3
-
Polymerization and Polymer ChemistryPolymer Chemistry
Free radical polymerizationH H H H
C C
HHmonomer
R +
free radical
R C C
H H
initiation
(ethylene)
R C C
H H
C C
H H+ R C C
H H
C C
H Hpropagation
Initiator: example benzoyl peroxide
R C C
H H
C C
HH
+ R C C
H H
C C
H H
propagation
dimer Initiator: example - benzoyl peroxide
H H H
Chapter 14 - 8
C
H
O O C
H
C
H
O2 R= 2
-
Chemistry and Structure of PolyethylenePolyethylene
Adapted from Fig. 14.1, Callister & Rethwisch 8e.Rethwisch 8e.
Note: polyethylene is a long-chain hydrocarbonparaffin wax for candles is short polyethylene
Chapter 14 - 9
- paraffin wax for candles is short polyethylene
-
Bulk or Commodity Polymers
Chapter 14 -10
-
Bulk or Commodity Polymers (cont)
Chapter 14 -11
-
Bulk or Commodity Polymers (cont)
Chapter 14 -12
-
VMSE: Polymer Repeat Unit Structures
Chapter 14 -13
Manipulate and rotate polymer structures in 3-dimensions
-
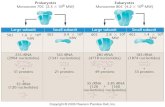
MOLECULAR WEIGHT Molecular weight, M: Mass of a mole of chains.
Low M
high Mhigh M
Not all chains in a polymer are of the same lengthNot all chains in a polymer are of the same length i.e., there is a distribution of molecular weights
Chapter 14 -14
-
MOLECULAR WEIGHT DISTRIBUTION
moleculesof#totalpolymer of wttotalnM
Adapted from Fig. 14.4, Callister & Rethwisch 8e.
molecules of #total
M n x MM n xi MiM w wi Mi
xi = number fraction of chains in size range iMi = mean (middle) molecular weight of size range i
Chapter 14 -15
wi = weight fraction of chains in size range i
-
Molecular Weight Calculation
Example: average mass of a classStudent Weight
mass (lb)1 104
What is the averageweight of the students in1 104
2 1163 140
this class:a) Based on the number
fraction of students in4 1435 1806 182
fraction of students in each mass range?
b) Based on the weight fraction of students in6 182
7 1918 220
fraction of students in each mass range?
Chapter 14 -16
9 22510 380
-
Molecular Weight Calculation (cont.)Solution: The first step is to sort the students into weight ranges.
Using 40 lb ranges gives the following table:
weight number of mean number weightrange students weight fraction fraction
Ni Wi xi wi
Calculate the number and weight fraction of students in each weight range as follows:
mass (lb) mass (lb)81-120 2 110 0.2 0.117121-160 2 142 0.2 0.150161 200 3 184 0 3 0 294
xi NiNi wi
NiWiNiWi
161-200 3 184 0.3 0.294201-240 2 223 0.2 0.237241-280 0 - 0 0.000281 320 0 0 0 000
For example: for the 81-120 lb range
x81120 210 0.2281-320 0 - 0 0.000321-360 0 - 0 0.000361-400 1 380 0.1 0.202
10
117.01881
011 x 212081 w
Chapter 14 -17
Ni NiWi10 1881
total number
total weight
-
Molecular Weight Calculation (cont.)weight mean number weightrange weight fraction fraction
Wi xi wii i imass (lb) mass (lb)81-120 110 0.2 0.117
121-160 142 0.2 0.150161-200 184 0.3 0.294201-240 223 0.2 0.237241-280 - 0 0.000281 320 0 0 000
281-320 - 0 0.000321-360 - 0 0.000361-400 380 0.1 0.202
Mn xiMi (0.2 x 110 0.2 x 142+ 0.3 x 184+ 0.2 x 223+ 0.1 x 380) =188 lbMw wiMi (0.117 x 110 0.150 x 142+ 0.294 x 184
Chapter 14 -18
+ 0.237 x 223+ 0.202 x 380) = 218 lb Mw wiMi 218 lb
-
Degree of Polymerization, DPDP = average number of repeat units per chain
C C C C C C C CH
H H H H H H H H
C C C C
H H H H
H( ) DP = 6H H H H H H H H H H H H
MDP n
m unitrepeatofweightmolecularaveragewherem
DP
mfm
m
:follows as calculated is this copolymers for
unitrepeatofweightmolecularaverage where
Chapter 14 -19
iimfm mol. wt of repeat unit iChain fraction
-
Molecular Structures for Polymers
secondarybonding
Adapted from Fig. 14.7, Callister & Rethwisch 8e.
Branched Cross-Linked NetworkLinear
Chapter 14 -20
-
Polymers Molecular ShapeMolecular Shape (or Conformation) chain
bending and twisting are possible by rotationbending and twisting are possible by rotation of carbon atoms around their chain bonds note: not necessary to break chain bondsnote: not necessary to break chain bonds
to alter molecular shape
Adapted from Fig. 14.5, Callister & Rethwisch 8e.
Chapter 14 -21
-
Chain End-to-End Distance, r
Adapted from Fig. 14.6, Callister & Rethwisch 8e.
Chapter 14 -22
-
Molecular Configurations for Polymers
Configurations to change must break bonds StereoisomerismStereoisomerism
C CHH
C C
H H
C C
H RC C
RHC C
H R
or C C
H H
E
A
C C
A
E
Stereoisomers are mirrorimages cant superimpose
ith t b ki b d EB D D B
E
mirror
without breaking a bond
Chapter 14 -23
plane
-
TacticityTacticity stereoregularity or spatial arrangement of R
units along chain
isotactic all R groups on same side of chain
syndiotactic R groups alternate sides
C C
H H HH
CC
HH
CC
HH
CC C C
H H
C C
H H
C C
H R RH
CCH R RH RH RH H R H RH H HH
Chapter 14 -24
-
Tacticity (cont.)
atactic R groups randomlypositionedpositioned
H H HH RH HH
C C
H R RH
CC
HH
CC
RH
CC
Chapter 14 -25
-
cis/trans Isomerism
C CHCH3
CH2 CH2C C
CH3
CH2
CH2
HCH2 CH2 CH2 H
cis transcis-isoprene
(natural rubber)
H t d CH
trans-isoprene (gutta percha)
H t d CH H atom and CH3 group on same side of chain
H atom and CH3 group on opposite sides of chain
Chapter 14 -26
-
VMSE: Stereo and Geometrical Isomers
Chapter 14 -27Chapter 7 - 19
Manipulate and rotate polymer structures in 3-dimensions
-
Copolymers Adapted from Fig. 14.9, Callister & R th i h 8
two or more monomers polymerized together
random
Rethwisch 8e.
random A and B randomly positioned along chain
alternating A and Balternating A and B alternate in polymer chain
block large blocks of A units alternate with large
alternating
units alternate with large blocks of B units
graft chains of B units f d A b kb
block
grafted onto A backbone
A B
Chapter 14 -28
A B graft
-
Crystallinity in Polymers Adapted from Fig. 14 10 C lli t &
Ordered atomic arrangements involving
14.10, Callister & Rethwisch 8e.
g gmolecular chains
Crystal structures in terms of nit cellsof unit cells
Example shownpolyethylene unit cell polyethylene unit cell
Chapter 14 -29
-
Polymer Crystallinity Crystalline regions
thin platelets with chain folds at facesthin platelets with chain folds at faces Chain folded structure
10 nm
Adapted from Fig. 14.12, Callister & Rethwisch 8e.
Chapter 14 -30
-
Polymer Crystallinity (cont.)Polymers rarely 100% crystalline Difficult for all regions of all chains to
become aligned
Degree of crystallinity
crystalline region
Degree of crystallinity expressed as % crystallinity.-- Some physical properties
depend on % crystallinity.depend on % crystallinity.-- Heat treating causes
crystalline regions to grow and % crystallinity to increase.
Adapted from Fig. 14.11, Callister 6e.
amorphousregion
Chapter 14 -31
p g ,(Fig. 14.11 is from H.W. Hayden, W.G. Moffatt,and J. Wulff, The Structure and Properties of Materials, Vol. III, Mechanical Behavior, John Wiley and Sons, Inc., 1965.)
-
Polymer Single Crystals Electron micrograph multilayered single crystals
(chain-folded layers) of polyethylene( y ) p y y Single crystals only for slow and carefully controlled
growth rates
Chapter 14 -32Adapted from Fig. 14.11, Callister & Rethwisch 8e.
-
Semicrystalline Polymers Some semicrystalline
polymers form yspherulite structures
Alternating chain-folded crystallites andcrystallites and amorphous regions
Spherulite structure for
Spherulite surface
prelatively rapid growth rates
surface
Adapted from Fig. 14.13, Callister & Rethwisch 8e.
Chapter 14 -33
-
Photomicrograph Spherulites in PolyethylenePolyethylene
Cross-polarized light used -- a maltese cross appears in each spherulite
Chapter 14 -34Adapted from Fig. 14.14, Callister & Rethwisch 8e.
-
ANNOUNCEMENTSReading:
Core Problems:
Self help Problems:Self-help Problems:
Chapter 14 -35