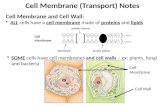
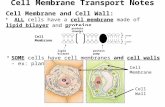
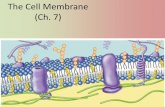
Cell Membrane The Door to the Cell. Structure of the Cell Membrane.
Cell membrane
-
Upload
mohammad-a-nassir -
Category
Health & Medicine
-
view
141 -
download
2
Transcript of Cell membrane
ORGANIZATION OF THE CELL
• cells consist of a nucleus surrounded by a nuclear membrane, and a cytoplasm surrounded by plasma membrane.
• cellular components (protoplasm) are made of :
1- water
2- Ions
3- proteins
4- lipids
5- carbohydrates
Cell membrane
• Thin, pliable and almost entirely made of proteins and lipids
the lipid bilayer impedes water penetrationcell membrane proteins are of two types:
integral and peripheral, these proteins perform various functions (channels, carriers, receptors, enzymes, controllers)
TRANSPORT THROUGH CELL MEMBRANE
• The lipid bilayer prevents mixing of ECF & ICF water and water-soluble substances cannot move freely through it.
• lipid-soluble substances move freely through the plasma membrane
• Water, ions and water-soluble substances are transported through proteins (channel proteins & carriers)
• Constant motion of molecules, ions, colloid particles through cell membrane
• Two types:1-Simple diffusion.2-facilitated.
diffusion
Simple diffusion
• Diffusion of a matter through the lipid bilayer is determined by its lipid solubility (O2, N2 ,CO2 move easily through the membrane)
• Water and some water-soluble substances (urea) pass through channels “pores” in proteins that penetrate all the way through the membrane
• Diffusion through channels is characterized by:
1-selective permeability (charge) , (size)
2-controled by opening and closing gates (voltage,
ligand)
• Carrier-mediated diffusion• the rate of diffusion reaches a maximum (Vmax)
• According to Fick’s law, the rate of diffusion depends on:
1-the magnitude of the concentration gradient2-the permeability of the plasma membrane to a substance.3-the surface area of the membrane across which diffusion takes place4-the molecular weight of a substance5-the distance through which diffusion takes place
Facilitated diffusion
Fig. 3-14a, p. 56
Step 1
Conformation X ofcarrier (binding sitesexposed to ECF)Molecule to betransported binds tocarrier
Molecule to betransported
Concentrationgradient
Plasmamembrane
Carrier molecule
(Low)
(High)ECF
ICF
Fig. 3-14b, p. 56
Step 2
On binding withmolecules to betransported, carrierchanges itsconformation
Conformation X of carrierConformation Yof carrier
Fig. 3-14c, p. 56
Step 3
Conformation Y ofcarrier (binding sitesexposed to ICF)Transported moleculedetaches from carrier
Direction oftransport
ECF
ICF
Fig. 3-14d, p. 56
Step 4
ECF
ICF
Conformation X ofcarrier (binding sitesexposed to ECF)After detachment,carrier reverts tooriginal shape
II. Active Transport:
Primary : - Secondary
Na-K pump Co- & counter-tr.
Ca - pump Na - Gl Na -aa
H - pump Na -H Na - Ca
Fig. 3-16, p. 58
Phosphorylatedconformation Yof carrier
Step 1
Phosphorylated conformation Y of carrier has high affinity for passenger. Molecule to be transported binds to carrier on low-concentration side.
Molecule to betransported Step 2
Dephosphorylated conformation X of carrier has low affinity for passenger. Transported molecule detaches from carrier on high-concentration side.
= phosphate
Direction oftransport
Concentrationgradient
(High)
(Low)
Dephosphorylatedconformation Xof carrier
ICF
ECF
Na+
Fig. 3-17, p. 59= Sodium (Na+) = Potassium (K+) = Phosphate
When open to the ECF, the carrier drops off Na+ on its high-concentration side and picks up K+ from its low-concentration side
Phosphorylated conformation Yof Na+–K+ pump has high affinityfor Na+ and low affinity for K+
when exposed to ICF
When open to the ICF, the carrier picks up Na+ from its low-concentration side and drops off K+ on its high-concentration side
Dephosphorylatedconformation X of Na+–K+
pump has high affinity forK+ and low affinity for Na+
when exposed to ECF
ICF
ECF
•Basic principles of osmosis: water diffuses from high [H2O] to region of lower [H2O] . (Kinetics).
Osmosis
OSMOTIC PRESSURE
•the exact amount of pressure required to stop osmosis.
•osmotic pressure is determined by the number of particles rather than their mass.
•osmole: 1 gram molecular weight of osmotically active solute
“ a solution having 1 osmole of solute dissolved in each Kg of water is said to have an osmolality of 1 osmole/Kg”• 1 milliosmole/liter 19.3 mmHg osmotic pressure at normal body temperature
Fig. 3-9, p. 53
100% water concentration 0% solute concentration
90% water concentration 10% solute concentration
= Water molecule = Solute molecule
Fig. 3-10, p. 53
Membrane
Higher H2Oconcentration,lower soluteconcentration
Lower H2Oconcentration,higher soluteconcentration
= Water molecule = Solute molecule
H2O
Fig. 3-11, p. 53
Membrane (permeable to both water and solute)
Side 1 Side 2
Higher H2O concentration,lower solute concentration
Lower H2O concentration,higher solute concentration
H2O moves from side 1 to side 2down its concentration gradient
Solute moves from side 2 to side 1down its concentration gradient
• Water concentrations equal• Solute concentrations equal• No further net diffusion• Steady state exists
Side 1 Side 2
= Water molecule
= Solute molecule
H2O
Solute
Fig. 3-12, p. 54
= Water molecule
= Solute molecule
Membrane (permeable to H2O but impermeable to solute)
Higher H2O concentration,lower solute concentration
Lower H2O concentration,higher solute concentration
H2O moves from side 1 to side 2down its concentration gradient
• Water concentrations equal• Solute concentrations equal• No further net diffusion• Steady state exists
Solute unable to move from side 2 toside 1 down its concentration gradient
Side 1 Side 2
Side 1 Side 2
Originallevel ofsolutions
H2O
Fig. 3-13, p. 54
= Water molecule
= Solute molecule
Membrane (permeable to H2O but impermeable to solute)
Pure water Lower H2O concentration,higher solute concentration
H2O moves from side 1 to side 2down its concentration gradient
Solute unable to move from side 2 toside 1 down its concentration gradient
Side 1 Side 2
Side 1 Side 2
Originallevel ofsolutions
H2O
• Water concentrations not equal• Solute concentrations not equal• Tendency for water to diffuse by
osmosis into side 2 is exactlybalanced by opposing tendency forhydrostatic pressure difference topush water into side 1
• Osmosis ceases• Opposing pressure necessary to
completely stop osmosis is equalto osmotic pressure of solution
Hydrostatic(fluid)pressuredifference
Osmosis
Hydrostatic pressure
1 osmole = 1 mole of solute particles (6.02 x10).
1 mole glucose = 1 osm. 1 mole NaCl = 2 osm. 1 mole Na2SO3 = 3 osm.
Relation between moles and osmoles
•Osmotic pressure : pressure that prevents the osmosis . •The higher the osmotic pressure of a solution, the lower its [H2O] but the higher its [solute]. •According to van’t hoff’s law:π = CRTπ= 19300 mm Hg for 1 osmole/liter at body temp.
π(osmotic pr.) C(solute con. In osmole/liter)R (ideal gas const.) T(absolute temp.)
• osmotic pr. = osmolarity(mOsm/L) X 19.3 mmHg
• the calculated value is not 100% correct due to intraionic
and intermolecular interactions between the particles and
it has to be multiplied by the “osmotic coefficient” of the
particles to reach the true value.
• the osmolarity of the body fluids is around 300 mOsm/L,
the plasma being 1mOsm/L higher because of the
osmotic effect of plasma proteins