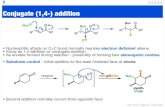
Catalyst 1.Draw the coolest picture of equilibrium you possibly can! Show off to your neighbor....
-
Upload
job-stewart -
Category
Documents
-
view
213 -
download
0
Transcript of Catalyst 1.Draw the coolest picture of equilibrium you possibly can! Show off to your neighbor....

Catalyst
1. Draw the coolest picture of equilibrium you possibly can! Show off to your neighbor.
2. What is a conjugate acid/base?
End

LECTURE 6.6.5 – BUFFERS (CONTINUED)

• 6.8 – I can identify the conjugate acid and base in a chemical reaction.
• 6.9 – I can explain what a buffer is, draw a titration curve for a buffered solution, and explain why these are essential to maintaining life in humans.
Today’s Learning Target

A Problem• Wendy Mata is an 10th grade student and she is
very nervous about her upcoming chemistry exam on acids and bases.
• Now Wendy, like many other people, begin to breath rapidly when she gets nervous.
• This is known as hyperventilation.

A Problem• Wendy begins to breath very rapidly.• The more she thinks of chemistry, the faster she
breaths, which causes her to think even more about chemistry.
• The world around Wendy begins to spin. She becomes very dizzy and passes out.

A Problem• An ambulance is quickly called.• When she gets to the hospital, the doctors give
her rapid treatment for her condition.• Wendy makes a full recovery and is able to take
her chemistry test.

Today’s Focus Question
• What caused Wendy to go into shock and how can this be prevented?

What are buffers?

I. Buffers• A buffer is a solution that resists
a change in pH.• Weak acids have both an acid
and a salt (that acts as a base) present at all times.– Same applies for weak bases
• Because both acid and base are present, these can react to prevent changes in pH

Example
CH3COO- + H2O CH3COOH + OH
CH3COO- + H+ CH3COOH
CH3COOH + OH- CH3COO- + H2O

Weak Acid and Strong Base Titration Curve
Buffering Region

Blood is a Buffer!• Human blood is buffered to have a pH of 7.3 – 7.5.• Carbonic acid in blood and acts as a buffer:
CO2 + H2O H2CO3 + HCO3-
• Acidosis is when blood becomes to acidic• Alkalosis is when blood becomes to basic

SUMMARIZE

You Are the Doctor!• You have the patient that was discussed on the
beginning of class.• With your group
(1) Develop a treatment to cure them if is acidosis
(2) Develop a treatment to cure them if it is alkalosis

Exit Ticket
1. Label the conjugate acid and base in the following reaction:
HCl + NH3 Cl- + NH4+
2. How do buffers prevent a change in a solutions pH?3. Draw a titration curve of a weak acid titrated with a strong base. Label the equivalence point and the buffering region.

Learning Log Assessment• Rate yourself 1 – 4 on LTs 6.8 and 6.9

Work Time• You have the remainder of the class period to
begin working on your formal lab report• The rough draft for this report will be due
Monday-Tuesday February 4th-5th


Closing Time• Test on Friday• Rough Draft of Lab Report due Tuesday

















![B.C.’S COOLEST TOWN [ ]](https://static.fdocuments.in/doc/165x107/61e74f15d0da670ff715534b/bcs-coolest-town-.jpg)