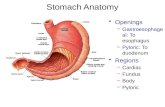
Ca stomach
-
Upload
radiation-oncology -
Category
Health & Medicine
-
view
336 -
download
0
Transcript of Ca stomach

MANAGEMENT OF CA STOMACH AND
ADJUVANT THERAPY

Symptoms and signs
diagnosis Treatment proper

Weight loss(21-60%)
Pain(62-91%)
Early satietyNausea and
vomiting(5-40%)
asymptomatic
commonest
Metastatic setting
GE JUNCTION
PYLORUS
Ascites, jaundice, or a palpable mass indicates incurable diseaseKrukenberg’ s tumor/Blumer’s shelf/ Sister Mary Joseph’s node/ Virchow’s node

TESTENDOSCOPY Direct visualization /cytology. Biopsy usual in 90% cases
But linitis plastica & small<3 cm & cardia lesion is difficult to diagnose
DOUBLE CONTRAST STUDY
: small lesion limited to inner layer of stomach wall.
CECT SCAN:
For both extent of spread & radiation portal (abdomen)Mediastinal LN ( in case of distal esophageal junction and thoracic mets.)
HELICAL CT:
More useful In detection of smaller LN
LAPAROSCOPIC STUDY:
Helps in detection in metastatic disease in case of operable lesion in preoperative imaging. Peritoneal fluid should be sampled in case of +ve is considered as M1 disease.
• T staging is accurate enough in 86 % case by EUS. Whereas 43% by CT.• EUS is 1st line imaging modality in T category• Diffuse /mucinous tumors – pet has lower detection rate. As FDG
accumulation is lower in this cases


Esopahgeal cancers and growth within 5 cm from GEJ are staged as Esophageal ca.All other ca having midpoint in stomach 5 cm distal to GEJ & within GEJ not involving the GEJ is termed as gastric ca.

Borrmann’s classification:Type 1: exophyticType 2: ulcerative lesion with elevated borders good prognosisType 3: ulcerstive lesion infiltrating stomach wall Type 4: diffusely infiltrating Type 5: unclassifiable.
PROGNOSTIC FACTORS:
• Tumor Extent Surrounding Tissue Involvement poor prognosis
• Pfs• Alk Phos Increase• Ethnicicty• Aneuploidy
Minimal node involvement does not alter prognosis.

TREATMENT

SURGERYPOTENTIALLY CURATIVE.
D1
D1+
D1
D2EMR
PERIGASTRIC LN
1-6 LN station.
COELIC LN
1-11 LN
station

EMR Created by Japanese research society for gastric caSince only mucosal involvement, nodal mets chance is 1%
• Diameter <3 cm • Easily manipulated area• Not done in
submucosal invasion as chances of nodal mets is high.
No RCT

Target –R0 resection
GASTRECTOMY
Partial total
• 5 cm margin on both side to have a R0 resection• recent concept about CRM(more incidence due to locally
advanced disease.)• Role of Frozen section
indicated for resectable stage IB–IIIDisease.

Mid and distal gastric cancer:• question is partial or total• On the basis of morbidity , mortality and oncologic out come partial is
prefered than total gastrectomy.• Three small RCT outcome is comparable in both approaches.
Proximal gastric cancer:• Esophagogastric Junction(siewart Type II/III) • Proximal Gastric Cancer
Options:• Transhiatal Esophagogatsrectomy (Cervical /Thoracic
Anastomosis)• Total Gastrectomy Transthoracic is better than transhiatal on basis of
perioperative morbidity 5 year survival better in TTEG(not statistically significant) No concensus in siewart type II/III Individualized on the basis of surgeon, age ,comorbidity,
PFS,T status, Nstatus

Lymphadenectomy:1. Adequate staging 2.Adequate therapyAt least 15 LN need to be retrieved.
Total gastrectomyD0: Lymphadenectomy less than D1D1: Nos. 1–7D1+:D1, Nos. 8a, 9, 11pD2: D1+Nos. 8a, 9, 10, 11p, 11d, 12a.
Distal gastrectomyD0: Lymphadenectomy less than D1D1: Nos. 1, 3, 4sb, 4d, 5, 6, 7D1+:D1,Nos. 8a, 9D2: D1+Nos. 8a, 9, 11p, 12a.
Japanese gastric cancer treatment guidelines 2010 (ver. 3)

Pylorus-preserving gastrectomyD0: Lymphadenectomy less than D1D1: Nos. 1, 3, 4sb, 4d, 6, 7D1+:D1,Nos. 8a, 9.
Proximal gastrectomyD0: Lymphadenectomy less than D1D1: Nos. 1, 2, 3a, 4sa, 4sb, 7D1+:D1,Nos. 8a, 9, 11p
Japanese gastric cancer treatment guidelines 2010 (ver. 3)

Type Descriptions
D1 lymphadenectomy T1a tumors that do not meet the criteria for EMR cT1bN0 tumors that are differentiated type and <1.5
cmD1+lymphadenectomy cT1N0 tumors other than the above
D2 lymphadenectomy potentially curable T2-T4 tumors, & cT1N+tumors. complete clearance of No. 10 nodes by splenectomy
should be considered for potentially curable T2-T4 tumors invading the greater curvature of the upper stomach.
D2+lymphadenectomy Non standard prophylactic para-aortic lymphadenectomy Denied by jcog 9501 prognosis of this population is poor.
Japanese gastric cancer treatment guidelines 2010 (ver. 3)
A recent meta-analysisof 12 randomised, controlled trials (RCTs) confirmed no overallsurvival (OS) benefit for D2 lymphadenectomy, although a benefit was seen among patients who had resection without a splenectomy and/or pancreatectomy

Adjuvant therapy:chemotherapy
1007 patientsStage II/III
tumor R0 D2
dissection
S-1
S-1 an oral fluoropyrimidine
(tegafur and oxonic acid)
Overall survival 80% vs. 70%
(improved)
Not sure about extrapolation to western population.

Meta analysis of 17 trials
Resectable gastric ca
post sx
Adjuvant CT
• Significant DFS, OS improvement with hazard ratio 0.82
• 5% improvement in 5 year survival.
• Conclusion: 5FU based CT is warranted.
Not sure about extrapolation to western population.


Closed early due to higher mortality rate in CTRT arm, but reaching to a plateau stage. Ultimately CTRT arm has superior
survival. Also that is more prominent in resected patients.

Adjuvant therapy:Radiothaerapy+/- chemotherapy
INT 0116Stage Ib –IV ca
N= 556
45GY/25#+
5FU leucovorin
Relapse free survival=48% vs.31%
(p<.001)
3 yr OS= 50% vs.41%
(p<.005)
OBS. Local recurrence 19% vs.29%
Clear survival advantage of chemoradiation strongly support its integration into the routine
care of patients with curatively resected high-risk carcinoma of the stomach and GE junction.

CALGB 80101 : POST OP CHEMO f/b CTRT f/b CT does not improve over all survival in comparison of ECF to CF
ARTIST studyKorean phase III study
458 patientsD2 resection
f/b chemoradiation (XRT+cepacitabine/cisplatin) vs.
Chemotherapy(capecitabine/cisaplatin)
• Not significant DFS• In subgroup analysis pathologic lymph node
superior DFS is foundConclusion: RT+CT does not significantly reduce
recurrence

ARTIST STUDY II
IS UNDER PROCESS
CRITICS trialStudy design:
Induction therapy (ECF) followed by D1+ resection followed by another 3 cycle +/-RT

PREOP CT/CTRT
Success of CTRT neoadjuvant setting in esophagus and rectum has raised enthusiasm in ca stomach which seems to be a logical approach.
Only phase II study are there regarding CTRT.Md Anderson cancer centre:
preop protocol of CDDP+leucovorin and 5FUf/b 45 GY /25# +5FU f/b D2 resection.
64 months longer median saurvival in pathological responding tumor than non responding(13%).
Another study showed FU+ paclitaxel+CDDP f/b 45 GY/25# +5 FU &leucovorin f/b D2 resection.
78% R0 resection, pathological complete response 25%, partial response 15%

PREOP CT/CTRT
POET study Induction therapy CDDP +5FU vs.
similar induction f/b concurrent etoposide /CDDP +RT
Preop CTRT arm- higher N0 rate.(64% vs.37%) pCR rate is high(16% vs. 2%)
Improved local control (76% vs. 59%) and overall survival.(47% vs. 28%)
Closed early due to slow accrual
Another TOPGEAR study is under process.

General Principles of Planning and Target Delineation for Adjuvant Radiation for Adenocarcinomas of the Gastro
esophageal Junction and the Stomach:
Fast for 2–3 h before CT simulation. Before treatments to ensure an
empty stomach and enhance daily treatment reproducibility
Planning CT scans of 3–5 mm thickness
Supine position with arms overhead, from top of the diaphragm (for stomach) or carina (for tumour of GE junction or cardia) to the bottom of L4.
Immobilisation with a Vac-Lok is recommended for treatment with IMRT
IV contrast is preferred to demonstrate blood vessels particularly for lymph nodes;
preoperative CT scans should be used to aid identification of preoperative tumour volume and nodal groups to be treated.
a total dose of 45 Gy in 25fractions of 1.8 Gy, 5 Fractions/week by 3D-conformal / intensity-modulated radiation therapy techniques

Target volumes
Definition and description
GTV Gross residual disease defined by CT imaging and surgical findings
PTV (residual disease)
GTV/positive margins + 1.5 cm. Cone down boost after 45 Gy to a total dose of 50.4 to 54Gy in 1.8Gy/fraction
CTV 45 Coverage of nodal groups according to subsite . Also includes remnant stomach, anastomosis (gastrojejunal, oesophagojejunal), duodenal stump
PTV 45 CTV 45+ 1 cm margin. A larger margin may be required for organ motion and setup uncertainties
Three areas must be identified as CTV for adjuvant radiotherapy: gastric tumour bed, anastomosis/ stumps regional lymphatics.
hepatogastric ligament should preferably be treated in all cases as it is at high risk of recurrence.
It represents the part of the lesser omentum that runs between the lesser curvature of the stomach and liver and contains the left and right gastric nodes that are not always completely removed at surgery





Advanced stage IV
CT
Single agent multiagent
Supportive care
Wagner et al . In a meta analysis of Cochrane collaboration found that overall survival is More in CT arm supporting evidence in favour of CT.(hazard ratio of 0.39)2 year survival is more in CT arm. QOL is also better in CT arm.
Wagner AD, Unverzagt S, Grothe W, et al . Chemotherapy for advanced cancer. CochraneDatabase Syst Rev 2010; (3):CD004064

Single Agent MultiagentS-1, 5fu, Capecitabine, Paclitaxel, docetaxel, irinotecan, Epi-/Doxorubicin.
Wagner et al. in their Cochrane review showed that multiagent is better than single agent.
Response rate ranges from 19%-49%Highest for S-1 and lowest For cisplatin & epi/doxorubicin.
OS is 8.3 moths vs. 6.7 months
HR is 0.82 Treatment related mortality
slightly higher in combination arm, but statistically significant.

CDDP+5FU
Established protocol decade long
Mostly used in control arm in various study
Losing its importance first as isolated use, with the advent of other agents.
KANG ET AL, REAL -2, FLAGS TRIAL showing oral formulation is non inferior than infusion.
DCF
TAX 325
Median TTP is 3.7 vs. 5.6 months
2 year survival is 8.8%vs. 18.4%
Response is 37%vs.28 %
Toxicity is also substantially increased.
*USFDA approval
FOLFIRI
Phase 2 trial proves advantage over folfiri vs. CF
PHASE 3 trial IF vs. CF objective response same . Toxicity somewhat less.

REAL -2 TRIAL
EOX
ECF
ECX
EOF
MEDIAN OS : ECF 9.9 months, EOF 9.3 months, ECX 9.9 months, and EOX 11.2 months The 1-year overall survival was also similar and ranged from 37.7% to
46.8%, the best outcome being seen with EOX and the lowest with the control arm of ECF . The authors concluded the oxal iplatin could be substituted for cisplatin, and capecitabine could be substituted for fluorouracil in the palliative setting.

TRANSTUZUMAB Overexpression or amplification of HER2 (EGFR2) 20 % patients with gastric
cancer. TOGA trial: median OS is 13.5 vs. 11.1 months. Response rate is 47 % vs. 35
%. Trastuzumab has been approved in Europe for HER2-positive gastric
cancer .
Targated therapyEGFR TRANSTUZUMAB,CETUXIMAB EGFR :TKI LAPATINIB,GEFTINIB,ERLOTINIBVEGF BEVACIZUMAB (AVAGAST trial)VEGF:TKI SUNITINIBmTOR inhibitor
EVEROLIMUS



THANK YOU