Biological Macromolecules Nature’s Building Blocks.
-
Upload
beryl-wilcox -
Category
Documents
-
view
217 -
download
0
Transcript of Biological Macromolecules Nature’s Building Blocks.

Biological Macromolecul
esNature’s Building Blocks

Create a way to put these in order.
Describe how you put them in order! - Particles of matter
- Atoms- Elements- Ecosystems- Biospheres
- Molecules- Macromolecules- Cell organelles- Cells- Galaxies
- The Universe - Tissues
- Organs- Organisms- Populations
- Planets- Planetary Systems with Stars

Prior Knowledge Discussion- Particles of matter- Atoms- Elements- Molecules- Macromolecules- Cell organelles- Cells- Tissues- Organs- Systems- Organisms- Populations- Ecosystems- Biospheres- Planets- Planetary Systems with Stars- Galaxies- The Universe
Smallest to largest!All of that is possible because of atoms!

Prior Knowledge Discussion
• Describe the characteristics required to be considered a living organism.
1. Composed of cells2. Reproduce3. Contain DNA4. Grow and Develop5. Use and obtain energy6. Respond to their environment7. Maintain a stable internal environment
/eliminate Waste8. Evolve: change over time

If everything is made of molecules,
what are molecules made of?
•Atoms!
Prior Knowledge Discussion

Which Elements are the Building Blocks of Life?
Almost all life forms on Earth are primarilymade up of only four basic elements:
• Carbon• Hydrogen• Oxygen• Nitrogen
(Phosphorous and Sulfur are also important in Biology)

Atoms are Elements!

Where are they Located on the Periodic
Table?

What are we Learning?Benchmark: SC.912L.18.1 Describe the basic molecular
structures and primary functions of the four major categories of biological macromolecules. AA

Definitions • Molecule: 2 or more atoms
chemically bonded togethero Example O2
• Compound: molecules that have more than 2 different elements chemically bonded togethero Example: H2O


Macromolecules are polymers
constructed of many organic
molecules called monomers.
• Monomer - small molecular subunit which joins (form covalent bonds) with similar units to form a polymer.
• Polymer - consists of up to millions of repeated, covalently linked monomers. It is a relatively small, simple molecule.

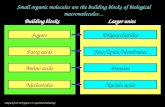
Life’s Macromolecules
• All key components of every living cell are made of macromolecules.
“Giant Molecules”• The four kinds of macromolecules:
1. Carbohydrates2. Lipids3. Proteins4. Nucleic acids

What you need to know
• For each macromolecule, you need to know:1. The elements it contains (building
blocks)2. The monomer structure (draw it)3. Important functions4. Examples
• Complete a Frayer-model for the 4 classes of macromolecules

Frayer Model
1. fold paper as demonstrated2. Label each tab 1 macromolecule3. Draw the structure of the macromolecule
on the front of each tab under the name4. On the inside:
o Building blockso Monomer nameo Functiono Exampleso *special notes

Building Blocks:
Composed of carbon (C), hydrogen (H), and oxygen (O) in a 1:2:1 ratio
Function:
Main source of energy for living things
Structural purposes
Examples:
Monosaccharaides like glucose, galactose, and fructose
Polysaccharides like glycogen and starch
cellulose in cell walls
Components:
Polysccharides or monosccharides (large or small units)
Carbohydrates
C6H12O
6

Carbohydrates (sugars)
• Composed of carbon (C), hydrogen (H), and oxygen (O) in a 1:2:1 ratioo Example: C6H12O6
• Made up of monomers called monosaccharide (simple sugars)

Carbohydrates• Basic structure: CnH2nOn
• Simple Carbs (monosaccharide) :o Glucoseo Fructoseo Galactoseo Ribose/deoxyribose
• Complex carbs (polysaccharide):o starch (bread, potatoes)o glycogen (beef muscle)o cellulose (lettuce, corn)

19
Carbohydrates
• Small sugar molecules to large sugar molecules.
• Examples:A. monosaccharideB. disaccharideC. polysaccharide
copyright cmassengale

20
Carbohydrates
Monosaccharide: one sugar unit
Examples: glucose (C6H12O6)
deoxyriboseriboseFructoseGalactose
glucose
copyright cmassengale

21
Carbohydrates
Disaccharide: two sugar unit
Examples: oSucrose
(glucose+fructose)oLactose
(glucose+galactose)oMaltose (glucose+glucose)glucoseglucose
copyright cmassengale

22
CarbohydratesPolysaccharide: many sugar
unitsExamples: starch (bread,
potatoes)glycogen (beef
muscle)cellulose
(lettuce, corn)
glucoseglucose
glucoseglucose
glucoseglucose
glucoseglucose
cellulose
copyright cmassengale

Building Blocks:
Mostly made from carbon and hydrogen atoms, some oxygen
Function:
The most important lipids are fats, which are energy storage molecules
Examples:
Fats, steroids, oils
Components:
A fat molecule consists of fatty acids joined to a molecule of glycerol = TRIGLYCERIDE (lipid monomer)
Lipids
Joined together by NON-POLAR COVALENT bonds

Glycerol Fatty Acid tail TRIGLYCERIDE
Monomers of Lipids are Triglycerides

ProteinsBuilding Blocks:
Proteins contain carbon, hydrogen, oxygen and nitrogen.
Functions: Control the rate of reactions
Regulate cell processes
Form bones and muscles
Transport substances into or out of cells
Help to fight disease
Examples:
ENZYMES—Speed up reaction rates
Components:
Composed of long chains of monomer subunits called amino acids.

Protein Structure
1.Amino group
2.R-group
3.Carboxyl group
Monomer: AMINO ACID:
There are 20 different amino acids

Nucleic Acids
Building Blocks: Containing carbon, hydrogen,
oxygen, nitrogen, and phosphorus.Function: Nucleic acids store and transmit
hereditary, or genetic information.
Examples: There are two types of nucleic
acids: DNA and RNA.

Nucleic AcidsComponents: Assembled from
individual monomers known as nucleotides.
Nucleotides consist of three parts: Five carbon sugar Phosphate group Nitrogenous base
Nucleotides (monomers) bond together to form DNA or RNA (polymer)

Making Polymers from Monomers
• CARBON can make 4 bonds with adjoining atoms
• When bonding two monomers together to make a polymer, DEHYDRATION SYNTHESIS occurso Removal of 2 Hydrogen's (H)
and 1 Oxygen (O) = - H2O


Breaking Polymers• The opposite reaction occurs when we
break the bond between monomers
• This is called a HYDROLYSIS reaction, because the addition of H2O will split the molecule’s covalent bond

Dehydration synthesis (condensation reaction) and Hydrolysis are reverse reactions

Building Macromolecules
Grab a lab packetGrab 2 partners (groups of 3)And listen for instructions