Biodiesel lesson 4a - Utah State · PDF file · 2012-03-09BIODIESEL Lesson 4a...
Transcript of Biodiesel lesson 4a - Utah State · PDF file · 2012-03-09BIODIESEL Lesson 4a...

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
BIODIESEL
Lesson 4a – Test biodiesel in an oil lamp

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
In a laboratory setting, test biodiesel in an oil lamp by estimating
calories and Btu.
• define density, energy density, calories and Btu
• calculate density, energy density, calories and Btu
• convert values between the units of calories and Btu and between volumetric and mass energy densities

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Fire Safety• Always follow directions.• Know where to locate and how to use the closest fire extinguisher.
• Do not use water to extinguish a fire involving biodiesel. Biodiesel is lighter than water, will float on the water, and spread the fire.
• Remember STOP, DROP, and ROLL!• Know evacuation plan in case of fire.

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
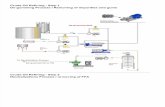
The Big Picture
• We want to find out how much energy biodiesel has compared to a standard value of diesel fuel.
• We will do this by burning the biodiesel to heat water. From the energy that is transferred into the water, we will be able to estimate how much energy is in the fuel.

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Steps• Step 1 – Find the density of the fuel.• Step 2 – Find the energy transferred from the burning fuel to the water.
• Step 3 – Find the mass energy density.• Step 4 – Convert to volumetric energy density.• Step 5 – Convert from metric values to standard values.
• Step 6 – Estimate the energy in biodiesel fuel• Step 7 – Estimate the energy ratio of biodiesel fuel to diesel fuel.

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Density• Density is a value of unit mass per unit volume.
• The Greek letter ρ (Rho) is often used to symbolize density.
• We will use the density of the liquid fuel to convert from a mass to a volumetric energy density.
• To find the density of a liquid, measure out a specific volume of the liquid and weigh it. Be sure that you do NOT include the weight of the container.
volume
mass=ρ

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Step 1 - Find the density of biodiesel
• If 21.0mL of biodiesel weigh 18.07g, what is its density?
mLgmL
g/860.0
0.21
07.18==ρ

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
The calorie and the Calorie
• 1 calorie (small c, abbreviated cal) is the energy it takes to raise 1 g of H2O by 1ºC.
• 1 Kilocalorie or Calorie (capital C, abbreviated kcal) is the energy it takes to raise 1 kg of H2O by 1ºC. It is equivalent to 1,000 calories (small c). This is also the unit of food Calories.

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Step 2 – Find the energy transferred from the burning fuel to the water
• Calculating the calories added to water (EH20) is a two step process.
1 – Calculate the temperature change (∆T) of the water by subtracting the initial temperature (Ti) from the final temperature (Tf).
2 – Multiply the mass of the water (MassH2O) by the temperature change.
• Use the equation:TxMassE OHOH ∆= 22
if TTT −=∆

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Calculating energy example
• If I have 247.4g of H2O and raise the temperature from 26.2ºC to 43.5ºC, how many calories were added to the H2O?
• ∆T = 43.5ºC-26.2ºC = 17.3ºC
• 247.4g x 17.3ºC = 4.28kgºC or 4.28kcal
• Or
kcalCxgE OH 28.4)2.265.46()4.247(2 =°−=

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Energy density• Energy density is different than density.
• Mass energy density is the amount of energy per unit mass.
• Volumetric energy density is the amount of energy per unit volume volume
energy
mass
energy
vol
mass
=
=
ρ
ρ

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Step 3 – Find the mass energy density
• If I burned 1.13g of biodiesel while adding 4.28 kilocalories to the H2O, how many calories were transferred into the water per gram of biodiesel?
g
kcal
g
kcalmass 79.3
13.1
28.4==ρ

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Step 4 – Convert to volumetric energy density
• Because of the nature of the burner, in our experiment we measured the mass of the fuel burned. However, since liquid fuels are purchased by volume, most liquid fuels are measured in volumetric energy density. Thus, we will convert from mass to volumetric energy density. This is done by using the following equation.
ρρρ xmassvol =

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Converting to volumetric energy density
• From step 1, biodiesel has 0.860g/mL and from step 3, 3.79kcal/g. How many calories are in 1 milliliter of biodiesel?
mL
kcal
mL
g
g
kcalvol 26.3860.0*79.3 ==ρ

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Energy Density
• Our energy density is then 3.79kcal/g or 3.26kcal/mL, depending on whether you desire a mass energy density or a volumetric energy density.

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
A Btu
• Since we live in the U.S.A., we use Standard units.
• A Btu is a British thermal unit• Similar to the calorie but is defined as the energy needed to raise 1 pound (lb) of water by 1ºF.
• We can use a direct conversion:• 1 Btu = 253 calories

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Step 5 – Convert from metric values to standard values
• To convert our metric volumetric energy density into a volumetric energy density using standard units, we need to make two conversions:
1 – convert kcal to Btu (253cal = 1Btu)
2 – convert mL to gal (3790 mL = 1 gal)
• Using the following formula, we can make both the conversions in the same equation.
gal
mLx
Btu
kcalmL
kcal
gal
Btu vol
vol 3790
253.0
ρρ =

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Simplify the conversion
• By combining the two constants in the previous equation, we can use the simplified equation below.
980,14xmL
kcal
gal
Btuvolvol ρρ =

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Energy density in Btu/gal
• From step 4, 3.26kcal/mL were transferred to the water. How many Btu/gal were transferred to the water?
gal
Btux
mL
kcal
gal
Btuvol 800,48980,1426.3 ==ρ

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Starting with English units
• The energy density can be calculated in Btu directly. Simply begin with English units and follow the same calculations.
• For mass energy density
• To convert to volumetric energy density
burnedbiodiesel
OH
massmass
Txmass
_
)()(2
∆=ρ
)()( ρρρ xmassvol =

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Why an estimate? – Loss to Environment
• The fuel actually contains more energy than what is transferred to the water.– air is also heated– flask is heated– fuel may not completely burn
• Due to the loss to the environment, the energy transferred to the water is less than the total energy contained in the fuel.
• It is impossible to stop loss to the environment and impractical in this lesson to calculate the loss. Air is heated
Glass is heated
Smoke indicates
Incomplete
combustion

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Why an estimate? – Energy transferred
• Another factor that affects our measurement is the energy transferred from the flame to the water. Several variables affect this value.– Flame size
– Distance from flame to flask
– Air flow
– Surrounding temperature
• In order to be consistent, as many of these factors should be kept constant as possible.

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Flame adjustment and soot• The larger the flame, the less complete the burn.
• The less complete the burn, the more soot the flame produces (after 15 min).

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Properly adjust the flame for consistency
• The flame should be adjusted to be about half blue flame and half (or a little less) yellow flame.
• Place it under the flask so that it is close enough to make the yellow flame disappear without extinguishing the flame entirely, which will happen if it is placed too close.
• The flame should be touching the flask.
• The distance between the flask and the burner should be kept constant from one test to another.

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Properly adjust the flame for consistency
• This is not the “only” way for the flame to be adjusted. The importance of adjusting the flame is to get consistency from measurements.
• It is OK to adjust the flame differently; just make sure that the flame is adjusted the same for each test.

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Energy density of petrodiesel• We can estimate the total value of Btu in biodiesel by making an estimate of how much energy we lose to the environment.
• Petrodiesel typically contains 128,700 Btu per US gallon.
• If petrodiesel is burned to heat water and 54,700 Btu per gallon is transferred to the water, then 43% (54,700/128,700) of the energy is transferred to the water and 57% of the energy is lost to the atmosphere. We will call the 57% our %loss.

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Assume similar loss• If we assume that the same percentage of energy will be lost to the environment when we burn the biodiesel, we can make an estimate of how much energy is in biodiesel.
• We start with two equations:
total
loss
lossOHtotal
E
Eloss
EEE
=
+=
%
2

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Calculate estimate
• Using algebra, we come up with the equation
• The total energy is equal to the energy transferred to the H2O (from step 5) divided by the percent transferred or one minus the percent lost to the environment (i.e. 38% = 0.38).
loss
EE OH
total%1
2
−=

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Step 6 – Estimate the energy in biodiesel fuel
• Using the previous equation, plug in the numbers to find an estimate of the energy density of biodiesel.
gal
Btugal
Btu
Etotal 000,11457.01
800,48
=−
=

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Step 7 – Estimate the energy ratio of biodiesel fuel to diesel fuel
• To find the energy ratio of our fuel to diesel fuel we divide the energy estimate by the value for diesel fuel.
• This appears to be a good estimate since the average energy value of biodiesel is about 91% the average energy value of diesel fuel per volume.
%89%100700,128
000,114=x

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Better methods
• This method is not very consistent and yields poor values when compared to other methods.
• However, this method of testing is inexpensive and easy to accomplish.
• To get accurate values, we would use equipment such as a pycnometer (to measure density) and a bomb calorimeter (to measure energy).

Biodiesel UnitBiodiesel UnitLesson 4a Lesson 4a –– Test biodiesel in an oil lampTest biodiesel in an oil lamp
Developed by Engineering and Technology Developed by Engineering and Technology Curriculum Team Curriculum Team ©©20082008—— www.etcurr.comwww.etcurr.com
Conclusion
• Now it is time for a demonstration!– Do Class Demonstration Worksheet 4a—Diesel Fuel.
• Burn biodiesel, not a tree!






![[Slideshare] fardh'ain-lesson#4a-contd-arkaan-ul-iiman-in-allah-(13 aprill2012)](https://static.fdocuments.in/doc/165x107/54ba38dc4a79595f5a8b4586/slideshare-fardhain-lesson4a-contd-arkaan-ul-iiman-in-allah-13-aprill2012.jpg)