ANODE AND CATHODE.docx
Transcript of ANODE AND CATHODE.docx
-
8/14/2019 ANODE AND CATHODE.docx
1/16
Here's a look at the difference between theanode and cathodeof a cell or battery and how youcan remember which is which.
Flow of Current
The anode and cathode are defined by the flow of current. In the general sense, current refers toany movement of electrical charge. However, you should keep in mind the convention that currentdirection is according to where a positivecharge would move, not a negative charge. So, ifelectrons do the actual movingin a cell, then current runs the opposite direction. Why is it definedthis way? Who knows, but that's the standard. Current flows in the same direction as positive
charge carriers, for example, when position ions carry the charge. Current flows opposite thedirection of negative charge carriers, such as electrons in metals.
Cathode
The cathode is the negatively charged electrode.
The cathode attracts cations or positive charge.
The cathode is the source of electrons or an electron donor. It may accept positive charge.
Because the cathode may generate electrons, which typically are the electrical species doing the actual movement, it may
be said that cathodes generate charge or that current moves from the cathode to the anode. This can be confusing,
because the direction of current would be defined by the way a positive charge would move. Just remember, any
movement of charged particles is current.
Anode
The anode is the positively charged electrode.
The anode attracts electrons oranions.
The anode may be a source of positive charge or an electron acceptor.
Cathode and Anode
Remember, charge can flow either from positive to negative or from negative to positive! Because
of this, the anode could be positively charged or negatively charged, depending on the situation.
The same is true for the cathode.
Keeping Them Straight
Remember the cathode attracts cations or the cathode attracts +charge. The anode
attractsnegative charge.
http://chemistry.about.com/od/chemistryterminology/a/Annode-Cathode-Difference.htmhttp://chemistry.about.com/od/chemistryterminology/a/Annode-Cathode-Difference.htmhttp://chemistry.about.com/od/chemistryterminology/a/Annode-Cathode-Difference.htmhttp://chemistry.about.com/od/chemistryglossary/a/Aniondefinition.htmhttp://chemistry.about.com/od/chemistryglossary/a/Aniondefinition.htmhttp://chemistry.about.com/od/chemistryglossary/a/Aniondefinition.htmhttp://chemistry.about.com/od/chemistryglossary/a/Aniondefinition.htmhttp://chemistry.about.com/od/chemistryterminology/a/Annode-Cathode-Difference.htm -
8/14/2019 ANODE AND CATHODE.docx
2/16
AnodeFrom Wikipedia, the free encyclopedia
This article needs additional citations for verification. Please
helpimprove this articlebyadding citations to reliable sources.
Unsourced material may bechallengedandremoved.(September 2012)
Diagram of azincanode in agalvanic cell.Note how electrons move out of the cell, and theconventional currentmoves
into it in the opposite direction.
An anodeis anelectrodethrough whichelectric currentflows intoa polarized electrical device. The
direction of electric current is, by convention, opposite to the direction of electron flow. In other words, the
electrons flow from the anode into, for example, anelectrical circuit.Mnemonic:ACID (Anode Current into
Device).
A widespread misconception is that anodepolarityis always positive (+). This is often incorrectly inferred
from the correct fact that in all electrochemical devices, negatively chargedanionsmove towards the anode
(hence their name) and positively chargedcationsmove away from it. In fact anode polarity depends on the
device type, and sometimes even in which mode it operates, as per the above electric current direction-
based universal definition. Consequently, as can be seen from the following examples, the anode is
positive in a device that consumes power, and the anode is negative in a device that provides power:
In a discharging battery orgalvanic cell(diagram at right), the anode is the negative terminal because
it is where the current flows into "the device" (i.e. the battery cell). This inward current is carried
externally by electrons moving outwards, negative charge moving one way constituting positive current
flowing the other way.
http://en.wikipedia.org/w/index.php?title=Anode&action=edithttp://en.wikipedia.org/w/index.php?title=Anode&action=edithttp://en.wikipedia.org/w/index.php?title=Anode&action=edithttp://en.wikipedia.org/wiki/Help:Introduction_to_referencing/1http://en.wikipedia.org/wiki/Help:Introduction_to_referencing/1http://en.wikipedia.org/wiki/Help:Introduction_to_referencing/1http://en.wikipedia.org/wiki/Template:Citation_neededhttp://en.wikipedia.org/wiki/Template:Citation_neededhttp://en.wikipedia.org/wiki/Template:Citation_neededhttp://en.wikipedia.org/wiki/Wikipedia:Verifiability#Burden_of_evidencehttp://en.wikipedia.org/wiki/Wikipedia:Verifiability#Burden_of_evidencehttp://en.wikipedia.org/wiki/Wikipedia:Verifiability#Burden_of_evidencehttp://en.wikipedia.org/wiki/Zinchttp://en.wikipedia.org/wiki/Zinchttp://en.wikipedia.org/wiki/Zinchttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Conventional_currenthttp://en.wikipedia.org/wiki/Conventional_currenthttp://en.wikipedia.org/wiki/Conventional_currenthttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electrical_circuithttp://en.wikipedia.org/wiki/Electrical_circuithttp://en.wikipedia.org/wiki/Electrical_circuithttp://en.wikipedia.org/wiki/Mnemonichttp://en.wikipedia.org/wiki/Mnemonichttp://en.wikipedia.org/wiki/Mnemonichttp://en.wikipedia.org/wiki/Electrical_polarityhttp://en.wikipedia.org/wiki/Electrical_polarityhttp://en.wikipedia.org/wiki/Electrical_polarityhttp://en.wikipedia.org/wiki/Anionhttp://en.wikipedia.org/wiki/Anionhttp://en.wikipedia.org/wiki/Anionhttp://en.wikipedia.org/wiki/Cationhttp://en.wikipedia.org/wiki/Cationhttp://en.wikipedia.org/wiki/Cationhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/File:Zinc_anode_2.svghttp://en.wikipedia.org/wiki/File:Zinc_anode_2.svghttp://en.wikipedia.org/wiki/File:Zinc_anode_2.svghttp://en.wikipedia.org/wiki/File:Zinc_anode_2.svghttp://en.wikipedia.org/wiki/File:Zinc_anode_2.svghttp://en.wikipedia.org/wiki/File:Zinc_anode_2.svghttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Cationhttp://en.wikipedia.org/wiki/Anionhttp://en.wikipedia.org/wiki/Electrical_polarityhttp://en.wikipedia.org/wiki/Mnemonichttp://en.wikipedia.org/wiki/Electrical_circuithttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Conventional_currenthttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Zinchttp://en.wikipedia.org/wiki/Wikipedia:Verifiability#Burden_of_evidencehttp://en.wikipedia.org/wiki/Template:Citation_neededhttp://en.wikipedia.org/wiki/Help:Introduction_to_referencing/1http://en.wikipedia.org/w/index.php?title=Anode&action=edit -
8/14/2019 ANODE AND CATHODE.docx
3/16
In a recharging battery, or anelectrolytic cell,the anode is the positive terminal, which receives current
from an external generator. The current through a recharging battery is opposite to the direction of
current during discharge; in other words, the electrode which was the cathode during battery discharge
becomes the anode while the battery is recharging.
In adiode,it is the positive terminal at the tail of the arrow symbol (base of the triangle), where current
flows into the device. Note electrode naming for diodes is always based on the direction of the forward
current (that of the arrow, in which the current flows "most easily"), even for types such as Zener
diodesor solar cells where the current of interest is the reverse current.
In acathode ray tube,it is the positive terminal where electrons flow out of the device, i.e., where
positive electric current flows in.
An electrode through which current flows the other way (out of the device) is termed acathode.
Contents
[hide]
1 Etymology
2 Flow of electrons
3 Electrolytic anode
4 Battery or galvanic cell anode
5 Vacuum tube anode
6 Diode anode
7 Sacrificial anode
8 Related antonym
9 See also
10 References
11 External links
Etymology[edit]
The word was coined in 1834 from theGreek (anodos), 'ascent', byWilliam Whewell,who had
been consulted[1]
byMichael Faradayover some new names needed to complete a paper on the recently
discovered process ofelectrolysis.In that paper Faraday explained that when an electrolytic cell is oriented
so that electric current traverses the "decomposing body" (electrolyte) in a direction "from East to West, or,
which will strengthen this help to the memory, that in which the sun appears to move", the anode is where
the current enters the electrolyte, on the East side: "anoupwards, odosa way ; the way which the sun
rises" (,[2]
reprinted in[3]
).
The use of 'East' to mean the 'in' direction (actually 'in' 'East' 'sunrise' 'up') may appear contrived.
Previously, as related in the first reference cited above, Faraday had used the more straightforward term
"eisode" (the doorway where the current enters). His motivation for changing it to something meaning 'the
http://en.wikipedia.org/wiki/Electrolytic_cellhttp://en.wikipedia.org/wiki/Electrolytic_cellhttp://en.wikipedia.org/wiki/Electrolytic_cellhttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Zener_diodehttp://en.wikipedia.org/wiki/Zener_diodehttp://en.wikipedia.org/wiki/Zener_diodehttp://en.wikipedia.org/wiki/Zener_diodehttp://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Cathodehttp://en.wikipedia.org/wiki/Cathodehttp://en.wikipedia.org/wiki/Cathodehttp://en.wikipedia.org/wiki/Anodehttp://en.wikipedia.org/wiki/Anodehttp://en.wikipedia.org/wiki/Anodehttp://en.wikipedia.org/wiki/Anode#Etymologyhttp://en.wikipedia.org/wiki/Anode#Etymologyhttp://en.wikipedia.org/wiki/Anode#Flow_of_electronshttp://en.wikipedia.org/wiki/Anode#Flow_of_electronshttp://en.wikipedia.org/wiki/Anode#Electrolytic_anodehttp://en.wikipedia.org/wiki/Anode#Electrolytic_anodehttp://en.wikipedia.org/wiki/Anode#Battery_or_galvanic_cell_anodehttp://en.wikipedia.org/wiki/Anode#Battery_or_galvanic_cell_anodehttp://en.wikipedia.org/wiki/Anode#Vacuum_tube_anodehttp://en.wikipedia.org/wiki/Anode#Vacuum_tube_anodehttp://en.wikipedia.org/wiki/Anode#Diode_anodehttp://en.wikipedia.org/wiki/Anode#Diode_anodehttp://en.wikipedia.org/wiki/Anode#Sacrificial_anodehttp://en.wikipedia.org/wiki/Anode#Sacrificial_anodehttp://en.wikipedia.org/wiki/Anode#Related_antonymhttp://en.wikipedia.org/wiki/Anode#Related_antonymhttp://en.wikipedia.org/wiki/Anode#See_alsohttp://en.wikipedia.org/wiki/Anode#See_alsohttp://en.wikipedia.org/wiki/Anode#Referenceshttp://en.wikipedia.org/wiki/Anode#Referenceshttp://en.wikipedia.org/wiki/Anode#External_linkshttp://en.wikipedia.org/wiki/Anode#External_linkshttp://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=1http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=1http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=1http://en.wikipedia.org/wiki/Greek_languagehttp://en.wikipedia.org/wiki/Greek_languagehttp://en.wikipedia.org/wiki/Greek_languagehttp://en.wikipedia.org/wiki/William_Whewellhttp://en.wikipedia.org/wiki/William_Whewellhttp://en.wikipedia.org/wiki/William_Whewellhttp://en.wikipedia.org/wiki/Anode#cite_note-1http://en.wikipedia.org/wiki/Anode#cite_note-1http://en.wikipedia.org/wiki/Michael_Faradayhttp://en.wikipedia.org/wiki/Michael_Faradayhttp://en.wikipedia.org/wiki/Michael_Faradayhttp://en.wikipedia.org/wiki/Electrolysishttp://en.wikipedia.org/wiki/Electrolysishttp://en.wikipedia.org/wiki/Electrolysishttp://en.wikipedia.org/wiki/Anode#cite_note-2http://en.wikipedia.org/wiki/Anode#cite_note-2http://en.wikipedia.org/wiki/Anode#cite_note-2http://en.wikipedia.org/wiki/Anode#cite_note-3http://en.wikipedia.org/wiki/Anode#cite_note-3http://en.wikipedia.org/wiki/Anode#cite_note-3http://en.wikipedia.org/wiki/Anode#cite_note-3http://en.wikipedia.org/wiki/Anode#cite_note-2http://en.wikipedia.org/wiki/Electrolysishttp://en.wikipedia.org/wiki/Michael_Faradayhttp://en.wikipedia.org/wiki/Anode#cite_note-1http://en.wikipedia.org/wiki/William_Whewellhttp://en.wikipedia.org/wiki/Greek_languagehttp://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=1http://en.wikipedia.org/wiki/Anode#External_linkshttp://en.wikipedia.org/wiki/Anode#Referenceshttp://en.wikipedia.org/wiki/Anode#See_alsohttp://en.wikipedia.org/wiki/Anode#Related_antonymhttp://en.wikipedia.org/wiki/Anode#Sacrificial_anodehttp://en.wikipedia.org/wiki/Anode#Diode_anodehttp://en.wikipedia.org/wiki/Anode#Vacuum_tube_anodehttp://en.wikipedia.org/wiki/Anode#Battery_or_galvanic_cell_anodehttp://en.wikipedia.org/wiki/Anode#Electrolytic_anodehttp://en.wikipedia.org/wiki/Anode#Flow_of_electronshttp://en.wikipedia.org/wiki/Anode#Etymologyhttp://en.wikipedia.org/wiki/Anodehttp://en.wikipedia.org/wiki/Cathodehttp://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Zener_diodehttp://en.wikipedia.org/wiki/Zener_diodehttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Electrolytic_cell -
8/14/2019 ANODE AND CATHODE.docx
4/16
East electrode' (other candidates had been "eastode", "oriode" and "anatolode") was to make it immune to
a possible later change in the direction convention forcurrent,whose exact nature was not known at the
time. The reference he used to this effect was the Earth's magnetic f ield direction, which at that time was
believed to be invariant. He fundamentally defined his arbitrary orientation for the cell as being that in which
the internal current would run parallel to and in the same direction as a hypothetical magnetizing current
looparound the local line of latitude which would induce a magnetic dipolefield oriented like the Earth's.
This made the internal current East to West as previously mentioned, but in the event of a later convention
change it would have become West to East, so that the East electrode would not have been the 'way in'
any more. Therefore "eisode" would have become inappropriate, whereas "anode" meaning 'East
electrode' would have remained correct with respect to the unchanged direction of the actual phenomenon
underlying the current, then unknown but, he thought, unambiguously defined by the magnetic reference. In
retrospect the name change was unfortunate, not only because the Greek roots alone do not reveal the
anode's function any more, but more importantly because, as we now know, the Earth's magnetic field
direction on which the "anode" term is based is subject toreversalswhereas thecurrentdirection
convention on which the "eisode" term was based has no reason to change in the future.
Since the later discovery of theelectron,an easier to remember, and more durably correct technically
although historically false, etymology has been suggested: anode, from the Greek anodos, 'way up', 'the
way (up) out of the cell (or other device) for electrons'.
Flow of electrons[edit]
The flow of electrons is always from anode to cathode outside of the cell or device, regardless of the cell or
device type and operating mode, with the exception of diodes, where electrode naming always assumes
current in the forward direction (that of the arrow symbol), i.e., electrons flow in the opposite direction, even
when the diode reverse-conducts either by accident (breakdown of a normal diode) or by design
(breakdown of a Zener diode, photo-current of a photodiode or solar cell).
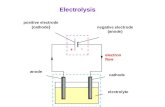
Electrolytic anode[edit]
Inelectrochemistry,the anodeis whereoxidationoccurs and is the positive polarity contact in
anelectrolytic cell.At the anode,anions(negative ions) are forced by the electrical potential to reactchemically and give off electrons (oxidation) which then flow up and into the driving circuit.Mnemonics:LEO
Red Cat (Loss of Electrons is Oxidation, Reduction occurs at the Cathode), or AnOx Red Cat (Anode
Oxidation, Reduction Cathode), or OIL RIG (Oxidation is Loss, Reduction is Gain of electrons), or Roman
Catholic and Orthodox (ReductionCathode, anodeOxidation), or LEO the lion says GER (Losing
electrons is Oxidation, Gaining electrons is Reduction).
This process is widely used in metals refining. For example, in copper refining, copper anodes, an
intermediate product from the furnaces, are electrolysed in an appropriate solution (such as sulfuric acid)to
yield high purity (99.99%) cathodes. Copper cathodes produced using this method are also described
aselectrolytic copper.
http://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Solenoidhttp://en.wikipedia.org/wiki/Solenoidhttp://en.wikipedia.org/wiki/Solenoidhttp://en.wikipedia.org/wiki/Solenoidhttp://en.wikipedia.org/wiki/Dipolehttp://en.wikipedia.org/wiki/Dipolehttp://en.wikipedia.org/wiki/Dipolehttp://en.wikipedia.org/wiki/Geomagnetic_reversalhttp://en.wikipedia.org/wiki/Geomagnetic_reversalhttp://en.wikipedia.org/wiki/Geomagnetic_reversalhttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=2http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=2http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=2http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=3http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=3http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=3http://en.wikipedia.org/wiki/Electrochemistryhttp://en.wikipedia.org/wiki/Electrochemistryhttp://en.wikipedia.org/wiki/Electrochemistryhttp://en.wikipedia.org/wiki/Oxidationhttp://en.wikipedia.org/wiki/Oxidationhttp://en.wikipedia.org/wiki/Oxidationhttp://en.wikipedia.org/wiki/Electrolytic_cellhttp://en.wikipedia.org/wiki/Electrolytic_cellhttp://en.wikipedia.org/wiki/Electrolytic_cellhttp://en.wikipedia.org/wiki/Anionhttp://en.wikipedia.org/wiki/Anionhttp://en.wikipedia.org/wiki/Anionhttp://en.wikipedia.org/wiki/List_of_chemistry_mnemonics#Electrodeshttp://en.wikipedia.org/wiki/List_of_chemistry_mnemonics#Electrodeshttp://en.wikipedia.org/wiki/List_of_chemistry_mnemonics#Electrodeshttp://en.wikipedia.org/wiki/Sulfuric_acidhttp://en.wikipedia.org/wiki/Sulfuric_acidhttp://en.wikipedia.org/wiki/Sulfuric_acidhttp://en.wikipedia.org/w/index.php?title=Electrolytic_copper.&action=edit&redlink=1http://en.wikipedia.org/w/index.php?title=Electrolytic_copper.&action=edit&redlink=1http://en.wikipedia.org/w/index.php?title=Electrolytic_copper.&action=edit&redlink=1http://en.wikipedia.org/w/index.php?title=Electrolytic_copper.&action=edit&redlink=1http://en.wikipedia.org/wiki/Sulfuric_acidhttp://en.wikipedia.org/wiki/List_of_chemistry_mnemonics#Electrodeshttp://en.wikipedia.org/wiki/Anionhttp://en.wikipedia.org/wiki/Electrolytic_cellhttp://en.wikipedia.org/wiki/Oxidationhttp://en.wikipedia.org/wiki/Electrochemistryhttp://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=3http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=2http://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Geomagnetic_reversalhttp://en.wikipedia.org/wiki/Dipolehttp://en.wikipedia.org/wiki/Solenoidhttp://en.wikipedia.org/wiki/Solenoidhttp://en.wikipedia.org/wiki/Electric_current -
8/14/2019 ANODE AND CATHODE.docx
5/16
Battery or galvanic cell anode[edit]
In abatteryorgalvanic cell,the anode is the negative electrode from which electrons flow out towards the
external part of the circuit. Internally the positively charged cations are flowing away from the anode (even
though it is negative and therefore would be expected to attract them, this is due toelectrode
potentialrelative to the electrolyte solution being different for the anode and cathode metal/electrolyte
systems); but, external to the cell in the circuit, electrons are being pushed out through the negative contact
and thus through the circuit by the voltage potential as would be expected. Note: in a galvanic cell, contrary
to what occurs in an electrolytic cell, no anions flow to the anode, the internal current being entirely
accounted for by the cations flowing away from it (cf drawing).
In the United States, many battery manufacturers regard the positive electrode as the anode, particularly in
their technical literature. Though technically incorrect, it does resolve the problem of which electrode is the
anode in a secondary (or rechargeable) cell. Using the traditional definition, the anode switches ends
between charge and discharge cycles.
Vacuum tube anode[edit]
In electronic vacuum devices such as acathode ray tube,the anode is the positively charged electron
collector. In a tube, the anode is a charged positive plate that collects the electrons emitted by the cathode
through electric attraction. It also accelerates the flow of these electrons.
Diode anode[edit]
In asemiconductordiode,the anode is the P-doped layer which initially suppliesholesto the junction. In
the junction region, the holes supplied by the anode combine with electrons supplied from the N-doped
region, creating a depleted zone. As the P-doped layer supplies holes to the depleted region, negative
dopant ions are left behind in the P-doped layer ('P' for positive charge-carrier ions). This creates a base
negative charge on the anode. When a positive voltage is applied to anode of the diode from the circuit,
moreholesare able to be transferred to the depleted region, and this causes the diode to become
conductive, allowing current to flow through the circuit. The terms anode and cathode should not be applied
to a Zener diode, since it allows flow in either direction, depending on the polarity of the applied potential
(i.e. voltage).
Sacrificial anode[edit]
http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=4http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=4http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=4http://en.wikipedia.org/wiki/Battery_(electricity)http://en.wikipedia.org/wiki/Battery_(electricity)http://en.wikipedia.org/wiki/Battery_(electricity)http://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Electrode_potentialhttp://en.wikipedia.org/wiki/Electrode_potentialhttp://en.wikipedia.org/wiki/Electrode_potentialhttp://en.wikipedia.org/wiki/Electrode_potentialhttp://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=5http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=5http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=5http://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=6http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=6http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=6http://en.wikipedia.org/wiki/Semiconductorhttp://en.wikipedia.org/wiki/Semiconductorhttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Electron_holehttp://en.wikipedia.org/wiki/Electron_holehttp://en.wikipedia.org/wiki/Electron_holehttp://en.wikipedia.org/wiki/Electron_holehttp://en.wikipedia.org/wiki/Electron_holehttp://en.wikipedia.org/wiki/Electron_holehttp://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=7http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=7http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=7http://en.wikipedia.org/wiki/File:Anodes-on-jacket.jpghttp://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=7http://en.wikipedia.org/wiki/Electron_holehttp://en.wikipedia.org/wiki/Electron_holehttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Semiconductorhttp://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=6http://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=5http://en.wikipedia.org/wiki/Electrode_potentialhttp://en.wikipedia.org/wiki/Electrode_potentialhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Battery_(electricity)http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=4 -
8/14/2019 ANODE AND CATHODE.docx
6/16
Sacrificial anodes mounted "on the fly" for corrosion protection of a metal structure
Incathodic protection,a metal anode that is more reactive to the corrosive environment of the system to be
protected is electrically linked to the protected system, and partiallycorrodesor dissolves, which protects
the metal of the system it is connected to. As an example, anironorsteelship's hullmay be protected by a
zincsacrificial anode,which will dissolve into the seawater and prevent the hull from being corroded.
Sacrificial anodes are particularly needed for systems where astatic chargeis generated by the action of
flowing liquids, such as pipelines and watercraft.
In 1824 to reduce the impact of this destructive electrolytic action on ships hulls, their fastenings and
underwater equipment, the Victorian scientist-engineer Sir Humphry Davy, developed the first and still most
widely used marine electrolysis protection system. Davy installed sacrificial anodes made from a more
electrically reactive (less noble) metal attached to the vessel hull and electrically connected to form a
cathodic protection circuit.
A less obvious example of this type of protection is the process ofgalvanisingiron. This process coats iron
structures (such as fencing) with a coating ofzincmetal. As long as the zinc remains intact, the iron is
protected from the effects of corrosion. Inevitably, the zinc coating becomes breached, either by cracking or
physical damage. Once this occurs, corrosive elements act as an electrolyte and the zinc/iron combination
as electrodes. The resultant current ensures that the zinc coating is sacrificed but that the base iron does
not corrode. Such a coating can protect an iron structure for a few decades, but once the protecting coating
is consumed, the iron rapidly corrodes.
If, conversely, tin is used to coat steel, when a breach of the coating occurs it actually accelerates oxidation
of the iron.
At least one anode is found in tank-type water heaters. The anode should be removed and checked after 5
years (sooner if there is a sodium based water softner inline), and replaced if 15 cm (6 inches) or more of
bare wire is showing. This will greatly extend the life of the tank.
Related antonym[edit]
The opposite of an anode is acathode.When the current through the device is reversed,
theelectrodesswitch functions, so anode becomes cathode, while cathode becomes anode, as long as the
reversed current is applied, with the exception of diodes where electrode naming is always based on the
forward current direction.
http://en.wikipedia.org/wiki/Cathodic_protectionhttp://en.wikipedia.org/wiki/Cathodic_protectionhttp://en.wikipedia.org/wiki/Cathodic_protectionhttp://en.wikipedia.org/wiki/Corrosionhttp://en.wikipedia.org/wiki/Corrosionhttp://en.wikipedia.org/wiki/Corrosionhttp://en.wikipedia.org/wiki/Ironhttp://en.wikipedia.org/wiki/Ironhttp://en.wikipedia.org/wiki/Ironhttp://en.wikipedia.org/wiki/Steelhttp://en.wikipedia.org/wiki/Steelhttp://en.wikipedia.org/wiki/Hull_(watercraft)http://en.wikipedia.org/wiki/Hull_(watercraft)http://en.wikipedia.org/wiki/Hull_(watercraft)http://en.wikipedia.org/wiki/Sacrificial_anodehttp://en.wikipedia.org/wiki/Sacrificial_anodehttp://en.wikipedia.org/wiki/Sacrificial_anodehttp://en.wikipedia.org/wiki/Electrostaticshttp://en.wikipedia.org/wiki/Electrostaticshttp://en.wikipedia.org/wiki/Galvanisehttp://en.wikipedia.org/wiki/Galvanisehttp://en.wikipedia.org/wiki/Galvanisehttp://en.wikipedia.org/wiki/Zinchttp://en.wikipedia.org/wiki/Zinchttp://en.wikipedia.org/wiki/Zinchttp://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=8http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=8http://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=8http://en.wikipedia.org/wiki/Cathodehttp://en.wikipedia.org/wiki/Cathodehttp://en.wikipedia.org/wiki/Cathodehttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/File:Anodes-on-jacket.jpghttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Cathodehttp://en.wikipedia.org/wiki/Anode?veaction=edit&vesection=8http://en.wikipedia.org/wiki/Zinchttp://en.wikipedia.org/wiki/Galvanisehttp://en.wikipedia.org/wiki/Electrostaticshttp://en.wikipedia.org/wiki/Sacrificial_anodehttp://en.wikipedia.org/wiki/Hull_(watercraft)http://en.wikipedia.org/wiki/Steelhttp://en.wikipedia.org/wiki/Ironhttp://en.wikipedia.org/wiki/Corrosionhttp://en.wikipedia.org/wiki/Cathodic_protection -
8/14/2019 ANODE AND CATHODE.docx
7/16
CathodeFrom Wikipedia, the free encyclopedia
Diagram of acoppercathode in agalvanic cell(e.g., a battery). A positive current iflows out of the cathode.
A cathodeis anelectrodethrough whichelectric currentflows out ofa polarized electrical device. The
direction of electric current is,by convention,opposite to the direction ofelectronflowthus, electrons are
considered to flow toward the cathode electrode while current flows away from it. This convention is
sometimes remembered using themnemonicCCD for cathode current departs.
Cathodepolarityis not always negative. Although positively chargedcationsalways move towards the
cathode (hence their name) and negatively chargedanionsmove away from it, cathode polarity depends
on the device type, and can even vary according to the operating mode. In a device which consumes
power, the cathode is negative, and in a device which provides power, the cathode is positive:
In a discharging battery or agalvanic cellthe cathode is the positive terminal since that is where the
current flows out of the device (see drawing). This outward current is carried internally by positive ions
moving from theelectrolyteto the positive cathode (chemical energy is responsible for this "uphill"
motion). It is continued externally by electrons moving inwards, negative charge moving one way
constituting positive current flowing the other way. For example, theDaniell galvanic cell's copper
electrode is the positive terminal and the cathode.
In a recharging battery, or anelectrolytic cell,the cathode is the negative terminal, from which current
returns back to the external generator. For example, reversing the current direction in a Daniell
galvanic cellwould produce an electrolytic cell,[1]
where the copper electrode is the positive terminal
and the anode.
http://en.wikipedia.org/wiki/Copperhttp://en.wikipedia.org/wiki/Copperhttp://en.wikipedia.org/wiki/Copperhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Conventional_currenthttp://en.wikipedia.org/wiki/Conventional_currenthttp://en.wikipedia.org/wiki/Conventional_currenthttp://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Mnemonichttp://en.wikipedia.org/wiki/Mnemonichttp://en.wikipedia.org/wiki/Mnemonichttp://en.wikipedia.org/wiki/Electrical_polarityhttp://en.wikipedia.org/wiki/Electrical_polarityhttp://en.wikipedia.org/wiki/Electrical_polarityhttp://en.wikipedia.org/wiki/Ionhttp://en.wikipedia.org/wiki/Ionhttp://en.wikipedia.org/wiki/Ionhttp://en.wikipedia.org/wiki/Ionhttp://en.wikipedia.org/wiki/Ionhttp://en.wikipedia.org/wiki/Ionhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Electrolytehttp://en.wikipedia.org/wiki/Electrolytehttp://en.wikipedia.org/wiki/Electrolytehttp://en.wikipedia.org/wiki/Daniell_cellhttp://en.wikipedia.org/wiki/Daniell_cellhttp://en.wikipedia.org/wiki/Daniell_cellhttp://en.wikipedia.org/wiki/Electrolytic_cellhttp://en.wikipedia.org/wiki/Electrolytic_cellhttp://en.wikipedia.org/wiki/Electrolytic_cellhttp://en.wikipedia.org/wiki/Daniell_cellhttp://en.wikipedia.org/wiki/Daniell_cellhttp://en.wikipedia.org/wiki/Daniell_cellhttp://en.wikipedia.org/wiki/Daniell_cellhttp://en.wikipedia.org/wiki/Cathode#cite_note-Reversed_Daniell_Cell-1http://en.wikipedia.org/wiki/Cathode#cite_note-Reversed_Daniell_Cell-1http://en.wikipedia.org/wiki/Cathode#cite_note-Reversed_Daniell_Cell-1http://en.wikipedia.org/wiki/File:Copper_cathode_2.svghttp://en.wikipedia.org/wiki/File:Copper_cathode_2.svghttp://en.wikipedia.org/wiki/File:Copper_cathode_2.svghttp://en.wikipedia.org/wiki/File:Copper_cathode_2.svghttp://en.wikipedia.org/wiki/Cathode#cite_note-Reversed_Daniell_Cell-1http://en.wikipedia.org/wiki/Daniell_cellhttp://en.wikipedia.org/wiki/Daniell_cellhttp://en.wikipedia.org/wiki/Electrolytic_cellhttp://en.wikipedia.org/wiki/Daniell_cellhttp://en.wikipedia.org/wiki/Electrolytehttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Ionhttp://en.wikipedia.org/wiki/Ionhttp://en.wikipedia.org/wiki/Electrical_polarityhttp://en.wikipedia.org/wiki/Mnemonichttp://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Conventional_currenthttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Copper -
8/14/2019 ANODE AND CATHODE.docx
8/16
In adiode,it is the negative terminal at the pointed end of the arrow symbol, where current flows out of
the device. Note: electrode naming for diodes is always based on the direction of the forward current
(that of the arrow, in which the current flows "most easily"), even for types such as Zener
diodesorsolar cellswhere the current of interest is the reverse current.
Invacuum tubes(includingcathode ray tubes)it is the negative terminal where electrons flow in from
the wiring and through the tube's near vacuum, constituting a positive current flowing out of the device.
An electrode through which current flows the other way (into the device) is termed ananode.
Contents
[hide]
1 Etymology
2 Flow of electrons
3 Chemistry cathode
o 3.1 Electrolytic cell
o 3.2 Galvanic cell
o 3.3 Electroplating metal cathode (a.k.a Electrolysis)
4 Electronics and physics cathode
o 4.1 Vacuum tubes
o 4.2 Cold cathodes and hot cathodes
o
4.3 Diodes
5 See also
6 References
7 External links
Etymology[edit]
The word was coined in 1834 from theGreek (kathodos), 'descent' or 'way down', byWilliam
Whewell,who had been consulted[2]
byMichael Faradayover some new names needed to complete a
paper on the recently discovered process of electrolysis. In that paper Faraday explained that when an
electrolytic cell is oriented so that electric current traverses the "decomposing body" (electrolyte) in a
direction "from East to West, or, which will strengthen this help to the memory, that in which the sun
appears to move", the cathode is where the current leaves the electrolyte, on the West side:
"katadownwards, `odosa way ; the way which the sun sets".[3][4]
The use of 'West' to mean the 'out' direction (actually 'out' 'West' 'sunset' 'down', i.e. 'out of view')
may appear unnecessarily contrived. Previously, as related in the first reference cited above, Faraday had
used the more straightforward term "exode" (the doorway where the current exits). His motivation for
changing it to something meaning 'the West electrode' (other candidates had been "westode", "occiode"
and "dysiode") was to make it immune to a possible later change in the direction convention forcurrent,
http://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Zener_diodehttp://en.wikipedia.org/wiki/Zener_diodehttp://en.wikipedia.org/wiki/Zener_diodehttp://en.wikipedia.org/wiki/Zener_diodehttp://en.wikipedia.org/wiki/Solar_cellhttp://en.wikipedia.org/wiki/Solar_cellhttp://en.wikipedia.org/wiki/Solar_cellhttp://en.wikipedia.org/wiki/Vacuum_tubehttp://en.wikipedia.org/wiki/Vacuum_tubehttp://en.wikipedia.org/wiki/Vacuum_tubehttp://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Anodehttp://en.wikipedia.org/wiki/Anodehttp://en.wikipedia.org/wiki/Anodehttp://en.wikipedia.org/wiki/Cathodehttp://en.wikipedia.org/wiki/Cathodehttp://en.wikipedia.org/wiki/Cathodehttp://en.wikipedia.org/wiki/Cathode#Etymologyhttp://en.wikipedia.org/wiki/Cathode#Etymologyhttp://en.wikipedia.org/wiki/Cathode#Flow_of_electronshttp://en.wikipedia.org/wiki/Cathode#Flow_of_electronshttp://en.wikipedia.org/wiki/Cathode#Chemistry_cathodehttp://en.wikipedia.org/wiki/Cathode#Chemistry_cathodehttp://en.wikipedia.org/wiki/Cathode#Electrolytic_cellhttp://en.wikipedia.org/wiki/Cathode#Electrolytic_cellhttp://en.wikipedia.org/wiki/Cathode#Galvanic_cellhttp://en.wikipedia.org/wiki/Cathode#Galvanic_cellhttp://en.wikipedia.org/wiki/Cathode#Electroplating_metal_cathode_.28a.k.a_Electrolysis.29http://en.wikipedia.org/wiki/Cathode#Electroplating_metal_cathode_.28a.k.a_Electrolysis.29http://en.wikipedia.org/wiki/Cathode#Electronics_and_physics_cathodehttp://en.wikipedia.org/wiki/Cathode#Electronics_and_physics_cathodehttp://en.wikipedia.org/wiki/Cathode#Vacuum_tubeshttp://en.wikipedia.org/wiki/Cathode#Vacuum_tubeshttp://en.wikipedia.org/wiki/Cathode#Cold_cathodes_and_hot_cathodeshttp://en.wikipedia.org/wiki/Cathode#Cold_cathodes_and_hot_cathodeshttp://en.wikipedia.org/wiki/Cathode#Diodeshttp://en.wikipedia.org/wiki/Cathode#Diodeshttp://en.wikipedia.org/wiki/Cathode#See_alsohttp://en.wikipedia.org/wiki/Cathode#See_alsohttp://en.wikipedia.org/wiki/Cathode#Referenceshttp://en.wikipedia.org/wiki/Cathode#Referenceshttp://en.wikipedia.org/wiki/Cathode#External_linkshttp://en.wikipedia.org/wiki/Cathode#External_linkshttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=1http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=1http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=1http://en.wikipedia.org/wiki/Greek_languagehttp://en.wikipedia.org/wiki/Greek_languagehttp://en.wikipedia.org/wiki/Greek_languagehttp://en.wikipedia.org/wiki/William_Whewellhttp://en.wikipedia.org/wiki/William_Whewellhttp://en.wikipedia.org/wiki/William_Whewellhttp://en.wikipedia.org/wiki/William_Whewellhttp://en.wikipedia.org/wiki/Cathode#cite_note-2http://en.wikipedia.org/wiki/Cathode#cite_note-2http://en.wikipedia.org/wiki/Cathode#cite_note-2http://en.wikipedia.org/wiki/Michael_Faradayhttp://en.wikipedia.org/wiki/Michael_Faradayhttp://en.wikipedia.org/wiki/Michael_Faradayhttp://en.wikipedia.org/wiki/Cathode#cite_note-3http://en.wikipedia.org/wiki/Cathode#cite_note-3http://en.wikipedia.org/wiki/Cathode#cite_note-3http://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Cathode#cite_note-3http://en.wikipedia.org/wiki/Cathode#cite_note-3http://en.wikipedia.org/wiki/Michael_Faradayhttp://en.wikipedia.org/wiki/Cathode#cite_note-2http://en.wikipedia.org/wiki/William_Whewellhttp://en.wikipedia.org/wiki/William_Whewellhttp://en.wikipedia.org/wiki/Greek_languagehttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=1http://en.wikipedia.org/wiki/Cathode#External_linkshttp://en.wikipedia.org/wiki/Cathode#Referenceshttp://en.wikipedia.org/wiki/Cathode#See_alsohttp://en.wikipedia.org/wiki/Cathode#Diodeshttp://en.wikipedia.org/wiki/Cathode#Cold_cathodes_and_hot_cathodeshttp://en.wikipedia.org/wiki/Cathode#Vacuum_tubeshttp://en.wikipedia.org/wiki/Cathode#Electronics_and_physics_cathodehttp://en.wikipedia.org/wiki/Cathode#Electroplating_metal_cathode_.28a.k.a_Electrolysis.29http://en.wikipedia.org/wiki/Cathode#Galvanic_cellhttp://en.wikipedia.org/wiki/Cathode#Electrolytic_cellhttp://en.wikipedia.org/wiki/Cathode#Chemistry_cathodehttp://en.wikipedia.org/wiki/Cathode#Flow_of_electronshttp://en.wikipedia.org/wiki/Cathode#Etymologyhttp://en.wikipedia.org/wiki/Cathodehttp://en.wikipedia.org/wiki/Anodehttp://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Vacuum_tubehttp://en.wikipedia.org/wiki/Solar_cellhttp://en.wikipedia.org/wiki/Zener_diodehttp://en.wikipedia.org/wiki/Zener_diodehttp://en.wikipedia.org/wiki/Diode -
8/14/2019 ANODE AND CATHODE.docx
9/16
whose exact nature was not known at the time. The reference he used to this effect was the Earth's
magnetic field direction, which at that time was believed to be invariant. He fundamentally defined his
arbitrary orientation for the cell as being that in which the internal current would run parallel to and in the
same direction as a hypotheticalmagnetizing current looparound the local line of latitude which would
induce a magneticdipolefield oriented like the Earth's. This made the internal current East to West as
previously mentioned, but in the event of a later convention change it would have become West to East, so
that the West electrode would not have been the 'way out' any more. Therefore "exode" would have
become inappropriate, whereas "cathode" meaning 'West electrode' would have remained correct with
respect to the unchanged direction of the actual phenomenon underlying the current, then unknown but, he
thought, unambiguously defined by the magnetic reference. In retrospect the name change was
unfortunate, not only because the Greek roots alone do not reveal the cathode's function any more, but
more importantly because, as we now know, the Earth's magnetic field direction on which the "cathode"
term is based is subject toreversalswhereas thecurrentdirection convention on which the "exode" term
was based has no reason to change in the future.
Since the later discovery of theelectron,an easier to remember, and more durably technically correct
(although historically false), etymology has been suggested: cathode, from the Greek kathodos, 'way
down', 'the way (down) into the cell (or other device) for electrons'.
Flow of electrons[edit]
The flow of electrons is almost always from anode to cathode outside of the cell or device, regardless of the
cell or device type and operating mode. An exception is when a diodereverse-conducts, either by accident
(breakdown of a normal diode) or by design (breakdown of a Zener diode, photo-current of a photodiode or
solar cell).
Chemistry cathode[edit]
Inchemistry,a cathodeis theelectrodeof anelectrochemical cellat whichreductionoccurs; a
usefulmnemonicto remember this is AnOx RedCat (Oxidation at the Anode = Reduction at the
Cathode).Another mnemonicis to note the cathode has a 'c', as does 'reduction'. Hence, reduction at the
cathode. The cathode can be negative like when the cell is electrolytic (where electrical energy provided tothe cell is being used for decomposing chemical compounds); or positive like when the cell is galvanic
(where chemical reactions are used for generating electrical energy). The cathode supplies electrons to the
positively charged cations which flow to it from the electrolyte (even if the cell is galvanic, i.e., when the
cathode is positive and therefore would be expected to repel the positively charged cations; this is due
toelectrode potentialrelative to the electrolyte solution being different for the anode and cathode
metal/electrolyte systems in agalvanic cell).
The cathodic current, inelectrochemistry,is the flow ofelectronsfrom the cathode interface to a species
in solution. The anodic currentis the flow of electrons into the anode from a species in solution.
Electrolytic cell[edit]
http://en.wikipedia.org/wiki/Solenoidhttp://en.wikipedia.org/wiki/Solenoidhttp://en.wikipedia.org/wiki/Solenoidhttp://en.wikipedia.org/wiki/Dipolehttp://en.wikipedia.org/wiki/Dipolehttp://en.wikipedia.org/wiki/Dipolehttp://en.wikipedia.org/wiki/Geomagnetic_reversalhttp://en.wikipedia.org/wiki/Geomagnetic_reversalhttp://en.wikipedia.org/wiki/Geomagnetic_reversalhttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=2http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=2http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=2http://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=3http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=3http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=3http://en.wikipedia.org/wiki/Chemistryhttp://en.wikipedia.org/wiki/Chemistryhttp://en.wikipedia.org/wiki/Chemistryhttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Electrochemical_cellhttp://en.wikipedia.org/wiki/Electrochemical_cellhttp://en.wikipedia.org/wiki/Electrochemical_cellhttp://en.wikipedia.org/wiki/Reduction_(chemistry)http://en.wikipedia.org/wiki/Reduction_(chemistry)http://en.wikipedia.org/wiki/Reduction_(chemistry)http://en.wikipedia.org/wiki/Mnemonichttp://en.wikipedia.org/wiki/Mnemonichttp://en.wikipedia.org/wiki/Mnemonichttp://en.wikipedia.org/wiki/List_of_chemistry_mnemonics#Electrodeshttp://en.wikipedia.org/wiki/List_of_chemistry_mnemonics#Electrodeshttp://en.wikipedia.org/wiki/List_of_chemistry_mnemonics#Electrodeshttp://en.wikipedia.org/wiki/Electrode_potentialhttp://en.wikipedia.org/wiki/Electrode_potentialhttp://en.wikipedia.org/wiki/Electrode_potentialhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Electrochemistryhttp://en.wikipedia.org/wiki/Electrochemistryhttp://en.wikipedia.org/wiki/Electrochemistryhttp://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=4http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=4http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=4http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=4http://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Electrochemistryhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Electrode_potentialhttp://en.wikipedia.org/wiki/List_of_chemistry_mnemonics#Electrodeshttp://en.wikipedia.org/wiki/Mnemonichttp://en.wikipedia.org/wiki/Reduction_(chemistry)http://en.wikipedia.org/wiki/Electrochemical_cellhttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Chemistryhttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=3http://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=2http://en.wikipedia.org/wiki/Electronhttp://en.wikipedia.org/wiki/Electric_currenthttp://en.wikipedia.org/wiki/Geomagnetic_reversalhttp://en.wikipedia.org/wiki/Dipolehttp://en.wikipedia.org/wiki/Solenoid -
8/14/2019 ANODE AND CATHODE.docx
10/16
In anelectrolytic cell,the cathode is where the negative polarity is applied to drive the cell. Common results
of reduction at the cathode are hydrogen gas or pure metal from metal ions. When discussing the relative
reducing power of two redox agents, the couple for generating the more reducing species is said to be
more "cathodic" with respect to the more easily reduced reagent.
Galvanic cell[edit]
In agalvanic cell,the cathode is where the positivepoleis connected to allow the circuit to be completed:
as the anode of the galvanic cell gives off electrons, they return from the circuit into the cell through the
cathode.
Electroplating metal cathode (a.k.a Electrolysis)[edit]
When metal ions are reduced from ionic solution, they form a pure metal surface on the cathode. Items to
be plated with pure metal are attached to and become part of the cathode in the electrolytic solution.
Electronics and physics cathode[edit]
Inphysicsorelectronics,a cathodeis an electrode that emits electrons into the device.
Vacuum tubes[edit]
In a vacuum tube or electronic vacuum system, the cathode emits free electrons. Electrons are extracted
from metal electrodes either by heating the electrode, causingthermionic emission,or by applying a strong
electric field and causingfield electron emission.Electrons can also be emitted from theelectrodesof
certain metals when light offrequencygreater than the threshold frequency falls on it. This effect is
calledphotoelectric emission.
Cold cathodes and hot cathodes[edit]
Cathodes used forfield electron emissionin vacuum tubes are calledcold cathodes.Heated electrodes
orhot cathodes,frequently calledfilaments,are much more common. Most radios and television sets prior
to the 1970s used filament-heated-cathode electron tubes for signal selection and processing; to this day, a
hot cathode forms the source of the electron beam(s) in cathode ray tubesin many television sets and
computer monitors. Hot electron emitters are also used as the electrodes influorescent lampsand in the
source tubes ofX-raymachines.
Diodes[edit]
In asemiconductordiode,the cathode is theNdopedlayer of thePN junctionwith a high density of free
electrons due to doping, and an equal density of fixed positive charges, which are the dopants that have
http://en.wikipedia.org/wiki/Electrolytic_cellhttp://en.wikipedia.org/wiki/Electrolytic_cellhttp://en.wikipedia.org/wiki/Electrolytic_cellhttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=5http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=5http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=5http://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Polarity_(physics)http://en.wikipedia.org/wiki/Polarity_(physics)http://en.wikipedia.org/wiki/Polarity_(physics)http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=6http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=6http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=6http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=7http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=7http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=7http://en.wikipedia.org/wiki/Physicshttp://en.wikipedia.org/wiki/Physicshttp://en.wikipedia.org/wiki/Physicshttp://en.wikipedia.org/wiki/Electronicshttp://en.wikipedia.org/wiki/Electronicshttp://en.wikipedia.org/wiki/Electronicshttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=8http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=8http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=8http://en.wikipedia.org/wiki/Thermionic_emissionhttp://en.wikipedia.org/wiki/Thermionic_emissionhttp://en.wikipedia.org/wiki/Thermionic_emissionhttp://en.wikipedia.org/wiki/Field_electron_emissionhttp://en.wikipedia.org/wiki/Field_electron_emissionhttp://en.wikipedia.org/wiki/Field_electron_emissionhttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Frequencyhttp://en.wikipedia.org/wiki/Frequencyhttp://en.wikipedia.org/wiki/Frequencyhttp://en.wikipedia.org/wiki/Photoelectric_emissionhttp://en.wikipedia.org/wiki/Photoelectric_emissionhttp://en.wikipedia.org/wiki/Photoelectric_emissionhttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=9http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=9http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=9http://en.wikipedia.org/wiki/Field_electron_emissionhttp://en.wikipedia.org/wiki/Field_electron_emissionhttp://en.wikipedia.org/wiki/Field_electron_emissionhttp://en.wikipedia.org/wiki/Cold_cathodehttp://en.wikipedia.org/wiki/Cold_cathodehttp://en.wikipedia.org/wiki/Cold_cathodehttp://en.wikipedia.org/wiki/Hot_cathodehttp://en.wikipedia.org/wiki/Hot_cathodehttp://en.wikipedia.org/wiki/Hot_cathodehttp://en.wiktionary.org/wiki/Filamentshttp://en.wiktionary.org/wiki/Filamentshttp://en.wiktionary.org/wiki/Filamentshttp://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wikipedia.org/wiki/Fluorescent_lamphttp://en.wikipedia.org/wiki/Fluorescent_lamphttp://en.wikipedia.org/wiki/Fluorescent_lamphttp://en.wikipedia.org/wiki/X-rayhttp://en.wikipedia.org/wiki/X-rayhttp://en.wikipedia.org/wiki/X-rayhttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=10http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=10http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=10http://en.wikipedia.org/wiki/Semiconductor_devicehttp://en.wikipedia.org/wiki/Semiconductor_devicehttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/w/index.php?title=N%E2%80%93doped&action=edit&redlink=1http://en.wikipedia.org/w/index.php?title=N%E2%80%93doped&action=edit&redlink=1http://en.wikipedia.org/w/index.php?title=N%E2%80%93doped&action=edit&redlink=1http://en.wikipedia.org/w/index.php?title=N%E2%80%93doped&action=edit&redlink=1http://en.wikipedia.org/w/index.php?title=N%E2%80%93doped&action=edit&redlink=1http://en.wikipedia.org/wiki/PN_junctionhttp://en.wikipedia.org/wiki/PN_junctionhttp://en.wikipedia.org/wiki/PN_junctionhttp://en.wikipedia.org/wiki/File:Diode_symbol.svghttp://en.wikipedia.org/wiki/File:Diode_symbol.svghttp://en.wikipedia.org/wiki/File:Diode_symbol.svghttp://en.wikipedia.org/wiki/File:Diode_symbol.svghttp://en.wikipedia.org/wiki/PN_junctionhttp://en.wikipedia.org/w/index.php?title=N%E2%80%93doped&action=edit&redlink=1http://en.wikipedia.org/wiki/Diodehttp://en.wikipedia.org/wiki/Semiconductor_devicehttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=10http://en.wikipedia.org/wiki/X-rayhttp://en.wikipedia.org/wiki/Fluorescent_lamphttp://en.wikipedia.org/wiki/Cathode_ray_tubehttp://en.wiktionary.org/wiki/Filamentshttp://en.wikipedia.org/wiki/Hot_cathodehttp://en.wikipedia.org/wiki/Cold_cathodehttp://en.wikipedia.org/wiki/Field_electron_emissionhttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=9http://en.wikipedia.org/wiki/Photoelectric_emissionhttp://en.wikipedia.org/wiki/Frequencyhttp://en.wikipedia.org/wiki/Electrodehttp://en.wikipedia.org/wiki/Field_electron_emissionhttp://en.wikipedia.org/wiki/Thermionic_emissionhttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=8http://en.wikipedia.org/wiki/Electronicshttp://en.wikipedia.org/wiki/Physicshttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=7http://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=6http://en.wikipedia.org/wiki/Polarity_(physics)http://en.wikipedia.org/wiki/Galvanic_cellhttp://en.wikipedia.org/wiki/Cathode?veaction=edit&vesection=5http://en.wikipedia.org/wiki/Electrolytic_cell -
8/14/2019 ANODE AND CATHODE.docx
11/16
-
8/14/2019 ANODE AND CATHODE.docx
12/16
Mnemonic Devices>Chemistry>Oxidation and Reduction (electrodes)
Dicarboxilic Acids (electrodes)
REDuction at CAThode: RED CAT
Oxidation and reduction in the electrodes of an electrochemical cell:
Oxidation takes place at the anode (both start with vowels), reductiontakes place at the cathode (both start with consonants).
REDuction at CAThode: RED CAT ANode for OXidation: AN OX
Reglas Mnemotcnicas>Quimica(es)>Oxidacion
http://www.ict4us.com/r.kuijt/index(en).htmhttp://www.ict4us.com/r.kuijt/index(en).htmhttp://www.ict4us.com/r.kuijt/index(en).htm#Chemistryhttp://www.ict4us.com/r.kuijt/index(en).htm#Chemistryhttp://www.ict4us.com/r.kuijt/index(en).htm#Chemistryhttp://www.ict4us.com/r.kuijt/en_oxidationreduction.htmhttp://www.ict4us.com/r.kuijt/en_oxidationreduction.htmhttp://www.ict4us.com/r.kuijt/en_oxidationreduction.htmhttp://www.ict4us.com/r.kuijt/index(es).htmhttp://www.ict4us.com/r.kuijt/index(es).htmhttp://www.ict4us.com/r.kuijt/index(es).htm#Quimica(es)http://www.ict4us.com/r.kuijt/index(es).htm#Quimica(es)http://www.ict4us.com/r.kuijt/index(es).htm#Quimica(es)http://www.ict4us.com/r.kuijt/es_oxidation.htmhttp://www.ict4us.com/r.kuijt/es_oxidation.htmhttp://www.ict4us.com/r.kuijt/es_oxidation.htmhttp://www.ict4us.com/r.kuijt/es_oxidation.htmhttp://www.ict4us.com/r.kuijt/index(es).htm#Quimica(es)http://www.ict4us.com/r.kuijt/index(es).htmhttp://www.ict4us.com/r.kuijt/en_oxidationreduction.htmhttp://www.ict4us.com/r.kuijt/index(en).htm#Chemistryhttp://www.ict4us.com/r.kuijt/index(en).htm -
8/14/2019 ANODE AND CATHODE.docx
13/16
Oxidacion
PIANO
en Pilas a la Izquierda el nodo , que es Negativo y
ocurre Oxidacin
Gracias Matthew
Reglas Mnemotcnicas>Quimica(es)>Las reacciones RedOx
Las reacciones RedOxPIANO:
Para recordar que en las reacciones RedOx,
al hacer el grfico de la Pila,
a la Izquierda se representa el nodo,
que es el que lleva el signo Negativo,
y donde se produce la Oxidacin.
Gracias Gene
Ezelsbruggetjes>Scheikunde>Hoe zit het met de kathode en anode?
http://www.ict4us.com/r.kuijt/index(es).htmhttp://www.ict4us.com/r.kuijt/index(es).htmhttp://www.ict4us.com/r.kuijt/index(es).htm#Quimica(es)http://www.ict4us.com/r.kuijt/index(es).htm#Quimica(es)http://www.ict4us.com/r.kuijt/index(es).htm#Quimica(es)http://www.ict4us.com/r.kuijt/es_piano.htmhttp://www.ict4us.com/r.kuijt/es_piano.htmhttp://www.ict4us.com/r.kuijt/es_piano.htmhttp://www.ict4us.com/r.kuijt/index.htmhttp://www.ict4us.com/r.kuijt/index.htmhttp://www.ict4us.com/r.kuijt/index.htm#Scheikundehttp://www.ict4us.com/r.kuijt/index.htm#Scheikundehttp://www.ict4us.com/r.kuijt/index.htm#Scheikundehttp://www.ict4us.com/r.kuijt/nl_elektrochemie.htmhttp://www.ict4us.com/r.kuijt/nl_elektrochemie.htmhttp://www.ict4us.com/r.kuijt/nl_elektrochemie.htmhttp://www.ict4us.com/r.kuijt/nl_elektrochemie.htmhttp://www.ict4us.com/r.kuijt/index.htm#Scheikundehttp://www.ict4us.com/r.kuijt/index.htmhttp://www.ict4us.com/r.kuijt/es_piano.htmhttp://www.ict4us.com/r.kuijt/index(es).htm#Quimica(es)http://www.ict4us.com/r.kuijt/index(es).htm -
8/14/2019 ANODE AND CATHODE.docx
14/16
Hoe zit het met de kathode en anode?
Hier een ezelsbruggetje voor bij elektrochemie
Oxiatie gebeurt aan deAnode
Reductie gebeurt aan deKathode
Het woordje is ORKA en als je de O en de A verbindt met een boogje en ookde k en de r krijg je iets dat uit de verte op een orka gelijkt.
Bron: Stijn
http://www.ict4us.com/r.kuijt/en_oxidationreduction.htm
http://www.ict4us.com/r.kuijt/en_oxidationreduction.htmhttp://www.ict4us.com/r.kuijt/en_oxidationreduction.htmhttp://www.ict4us.com/r.kuijt/en_oxidationreduction.htm -
8/14/2019 ANODE AND CATHODE.docx
15/16
Cations and Anions
Cations are pawsitive.
Cations are positive, while anions are negative:
The letter tin cation looks like a positive plus sign +: ca+ion Cations are pawsitive. An ANIONis A Negative ION
http://www.ict4us.com/mnemonics/en_cationsanions.htm
http://www.ict4us.com/mnemonics/en_cationsanions.htmhttp://www.ict4us.com/mnemonics/en_cationsanions.htmhttp://www.ict4us.com/mnemonics/en_cationsanions.htm -
8/14/2019 ANODE AND CATHODE.docx
16/16
Oxidation and Reduction (electrons)
Acronyms for oxidation and reduction:
Oxidation Is Losing Electrons, Reduction Is Gaining Electrons: OIL RIG Electron Loss Means Oxidation: ELMO Losing Electrons Oxidation, Gaining Electrons Reduction: LEOthe
lion. GER! or LEOsays GER
http://www.ict4us.com/mnemonics/en_oxidation_reduction.htm
http://www.ict4us.com/mnemonics/en_oxidation_reduction.htmhttp://www.ict4us.com/mnemonics/en_oxidation_reduction.htmhttp://www.ict4us.com/mnemonics/en_oxidation_reduction.htm