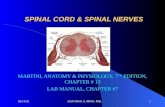
Anatomy of the Spinal Cord Structure of the spinal cord Tracts of the spinal cord
An Evidence-based Pressure Ulcer Monitoring Tool for Spinal Cord Injury/Disease (SCI-PUMT)
-
Upload
libby-perkins -
Category
Documents
-
view
23 -
download
0
description
Transcript of An Evidence-based Pressure Ulcer Monitoring Tool for Spinal Cord Injury/Disease (SCI-PUMT)

An Evidence-based Pressure Ulcer Monitoring Tool for Spinal Cord Injury/Disease (SCI-PUMT)Gail Powell-Cope PhD, ARNP, FAANActing Director, HSR&D/RR&D Center of ExcellenceTampa, [email protected]

Monitoring Pressure Ulcer Healing in Persons with Spinal Cord Impairment
Funded by VA Health Services Research and
Development Service (HSR&D)
Nursing Research Initiative 03-245, IRB#:
104145, 2006 – 2008
These findings and conclusions do not
necessarily represent the
Department of Veteran Affairs or HSR&D

Investigators
Co-Principal Investigators
◦Susan S. Thomason DNP, MN, RN
◦Audrey Nelson PhD, RN, FAAN
(Retired)
Co-Investigators
◦Steven Luther PhD
◦Jeffrey J. Harrow MD, PhD, FACP

Study StaffPolly Placios, MS (Project/Data
Manager)Data Collectors
◦Stephanie McGovern, RN◦Francis Hernandez, RN◦Suk Tomlinson, RN◦Olivia Monteso-Smithson, RN◦Linda Smith, RN
Mary Reeder, BIS (Program Assistant)

Conclusion
This study found that the SCI-PUMT was a reliable, valid, and sensitive
instrument for measuring PrU healing in persons with SCI in a
100 bed VHA SCI/D Center.

“Problems” (or challenges)Clinical Problem
◦Pressure ulcers are a high volume, high cost condition in Spinal Cord Impairment
Implementation◦Translating consistent and quality
pressure ulcer monitoring into clinical practice, across all 32 SCI/D Centers, is a challenge. It takes 17 years from for new knowledge
generated by a randomized controlled trial to be incorporated into practice, and even then, the application is highly uneven (Balas & Boren, 2000).


Options for Implementing Changes
1. Dissemination Alone (journal articles, distribution of printed materials, CME) (not effective)
2. Educational Outreach (Academic Detailing and Local Opinion Leaders) (promising and mixed evidence)
3. Computer-based decision support systems (mixed)
4. Audit and Feedback (mixed evidence)
5. Patient-mediated Interventions such as education, reminders (promising)
6. Patient-specific clinical reminders (promising)

PARiHS FrameworkPromoting Action on Research
Implementation in Health Services
Successful implementation is a function of: ◦the nature and type of evidence ◦qualities of the context in which the
evidence is being introduced, and ◦the way implementation is facilitated

Evidence (Strong)
Evidence(Weak)
Context(Strong)
Context(Weak)
Ideal Situation for
Implementation
Innovation
Kitson, A. L., et al., (2008). Evaluating the successful implementation of evidence into practice using the PARiHS framework: theoretical and practical challenges. Implementation Science: IS, 3, 1. doi:10.1186/1748-5908-3-1
Innovation
Innovation
Faci
litat
ion
Facilit
ation
Facilitation

Clinical ProblemPersons with spinal cord impairment
(SCI) are at extreme risk for PrU due to immobility, lack of sensation, collagen degradation, moisture, nutritional status, transfers, decreased ability to self-perform pressure redistribution, pain, and other risk factors.
PrU prevalence is 14-32%. PrU affect morbidity, mortality,
function, quality of life, and economics.

Clinical Practice Guidelines
Consortium for Spinal Cord Medicine (2000) Recommendations:Modify the treatment plan if the
ulcer shows no evidence of healing within 2-4 weeks.
Evaluate healing progress using an instrument or other quantitative measurements.

Clinical Practice Guidelines
National Pressure Ulcer Advisory Panel (NPUAP) European Pressure Ulcer Advisory Panel (EPUAP) (2009) Recommendations:Assess progress toward healing…use a
validated tool…Re-evaluate the PrU, the plan of care,
and individual if the PrU does not show progress toward healing within 2 weeks…

SettingMichael Bilirakis
Spinal Cord Injury/Disorders Center
James A. Haley Veterans HospitalTampa, Florida
100 inpatient beds CARF-accredited)Large outpatient patient populationHome Care (CARF-accredited)Long Term Care

However…Variations in how PrU healing is
measured varies across sites.◦Bates-Jensen Wound Assessment Tool
(BWAT)◦Pressure Ulcer Scale for Healing (PUSH)◦Hybrid tools with little psychometric
evaluationThese variations limit the ability to
conduct comparable trials of interventions

Research Questions
1. Is the SCI-PUMT valid for measuring PrU healing?
2. Is the SCI-PUMT reliable for measuring PrU healing?
3. How sensitive is the SCI-PUMT for measuring healing over time?

SCI-PUMT Phases
I. Development of item pool
II. Development and testing of SCI-PUMT
III. Analysis and SCI-PUMT refinement
IV. Assessment of SCI-PUMT reliability

DEVELOPMENT OF ITEM POOL
Phase 1

Development of Item PoolExpert Panel #1
◦ Aim: Identify measures and variables important and/or specific to PrU healing in SCI population
◦ 1 day on-site, Tampa, Florida◦ 9 interdisciplinary experts (MDs, RNs, OT, PT,
RD)◦ Variables then sent to EP for comment
Expert Panel #2
◦ Aim: Obtain content validity (all relevant concepts) for item pool
◦ 11 interdisciplinary experts (MDs, RNs, RD)◦ Aggregated variables sent to EP for comment

Item Pool
Consisted of 30 items◦Items from two established PrU
healing assessment tools (PUSH, BWAT)
◦Additional items identified by Expert Panels

DEVELOPMENT AND TESTING OF SCI-PUMT
Phase II

Subjects
Recruited from Inpatient, Outpatient, Home Care
3-year longitudinal cohort study
Assessed 30 PrU variablesPrU unit of analysis

Inclusion CriteriaEnrolled in SCI/D Registry and
receiving primary care from JAHVA SCI primary physician
Primary or secondary diagnosis of
Stages II-IV PrUSCI duration more than 12
months

Exclusion Criteria
Immune compromised
Severely mentally ill or cognitively impaired
Terminally ill

Subject Profile
Sample Size 66 Unique Patients167 Pressure Ulcers
Age 60 years (mean)
Gender Male 98%
Level of Injury Tetra 49%; Para 46%
ASIA A 58%; B 20%; Other 23%
Years since SCI onset
23 Years (mean)
High School Graduate
80%

Pressure Ulcer Characteristic
Findings
Number PrU / subject 1 – 9 (mean 2.5)
Previous PrU 77%
Prior PU surgery 53%
Location Ischia 43% Sacrococcygeal 26% Trochanter 8%Heel 8%
Stage II 20%; III 38%; IV 42%
Ulcer Pain 18%
Chronic Osteomyelitis 33%

Co-Morbidity Incidence
Diabetes Mellitus 26%
Anemia 24%
Peripheral Vascular Disease
11%
Chronic Obstructive Pulmonary Disease
9%
Congestive Heart Failure
8%
Heterotopic Ossification
8%

Other Baseline Factors
Incidence
Immunosuppressant Medications
12%
Spasticity Interference with Function(1 = none; 5 = maximum)
2.4 mean (SD 1.6)
SpasticityModified Ashworth Scale(1 = slight ↑ tone; 5 =
rigidity)
2.1 mean (SD 1.2)
Pain(0 = none; 10 = severe)
3.9 (SD = 2.6)
Mean Body Weight 175 lb (SD 36.4)
Nicotine Preceding WeekSubstance Abuse
29%6%

Data Collection6 Registered Nurse Data Collectors13 time points: 30 variables +
VeV PhotographBaseline and 12 weeks or
◦Complete healing◦Patient withdrawal◦Hospital discharged and lived >40
miles◦Plastic surgery intervention

VeV Measurement Documentation Software
Digital images used to calculate:• Volume

Intra- and Inter-Rater Reliability Ranges4 RN Data CollectorsIntra-Rater Reliability1 DCSame PrUTwice 1 ½ hours
apart
Inter-Rater Reliability4 DCSame PrUConsecutively
TOOL Intra-RaterICC
Inter-Rater ICC
PUSH 0.880.996
0.76 – 0.96
BWAT 0.87 – 0.99
0.69 – 0.91

ANALYSIS AND SCI-PUMT REFINEMENT
Phase III

Statistical Analysis
Construct validity
Predictive validity
Sensitivity to change
Internal consistency reliability

Construct ValidityExploratory Factor Analysis (EFA)
N = 167 PrUPrincipal factor extraction with
Promax (orthogonal rotation)Items removed from analysis based
on: ◦Values in correlation matrix◦Factor loadings of similar items (from 2
tools)◦Items not well defined by factors (low
communalities)

Variable Source
Geometric Factor
Substance
Factor Depth PUMT .82 -
Tunneling PUMT .77 -
Edges PSST .55 -
Undermining PUMT .48
Surface area PUSH .35 .51
Necrotic amount PSST - .52
Exudate type PUMT - .40
* Factor loading < |.30| have been replaced with “-“for ease of reading
Factor Analysis Results

Predictive ValidityOutcome variables to represent
PrU healing: Surface Area & Volume
Criterion Validity - VeV MD Software (within limits)
Correlates with the gold standard
Regression analyses – SCI-PUMT at baseline

Predictive ValidityExplains outcome variationsDependent variable:
Volume (VeV Camera)Predictor variables: Factor
analysis itemsSCI PUMT explained an
estimated59% of the variance in volume over the course of the study

Comparison of Scales:Volume (by VeV) Regression
SCI-PUMT
PUSH(Pressure
Ulcer Scale for Healing)
BWAT (Bates-Jensen Wound
Assessment Tool)
R2
(estimated based on proportional reduction in mean squared prediction error as per Snijders &
Bosker, 1994)
59% 57% 24%

ASSESSMENT OF SCI-PUMT RELIABILITY
Phase IV

Internal Consistency Reliability
Cronbach’s alpha = 0.74 (using study data)

SCI-PUMT ReliabilityAim: Evaluate intra- and inter-
rater reliability of in a clinical setting
26 Nurses trained in SCI-PUMT at Tampa VA SCI/D Center
Two months later, two sets of 3 SCI RNs evaluated 16 ulcers twice with an interval of 1½ hours between assessments

ResultsClinician Reliability
Intra-rater reliability 0.81 – 0.99
Inter-rater reliability 0.79
All reliability measures found to be above our established acceptability threshold

VARIABLES AND SCORING
SCI-PUMT

Pressure Ulcer
Site:
Sacrum or
Coccyx
Trochanter
Ischium
Heel
Other
______
Body Side: Right
Left
Midline
Orientation:
Medial Lateral
Positioning
Upper Leg Flexed When
Turned:
Yes
No
Surface Turned Onto: Right Left Back
Abdomen
Spinal Cord Impairment Pressure Ulcer Monitoring Tool (SCI-PUMT)
Patient ___________________SS#______________________ Ulcer # ______

Variable Score Options Score Geometric Factor
Surface Area(L x W)
1 2 3 4 5<1 cm2 >1 - <2.5 cm2 >2.5 - <5 cm2 >5 - <10 cm2 >10 - <15 cm2
6 7 8 9 10>15 - <25 cm2 >25 - <35 cm2 >35 - <55 cm2 >55 - <85 cm2 >85 cm2
Depth 0 1 2 3 40 cm >0 - <1 cm >1 - <2 cm >2 - <3 cm >3 cm
Edges 1 Indistinct, diffuse, none
clearly visible Distinct, outline clearly
visible, attached, even with ulcer base
Well-defined, not attached to ulcer base
2 Well-defined, not attached to base, rolled
under, thickened Well-defined, fibrotic, scarred, or hyperkeratotic
Tunneling 0None
1≤ 2 cm
2> 2 - ≤ 4 cm
3>4 cm
Undermining 0None
1≤ 2 cm
2> 2 - ≤ 4 cm
3>4 cm
Sub-total Score Geometric Factor
Substance Factor
Exudate Type 0None
1Serous/Sanguineous
2Green/Purulent
Necrotic Tissue Amount 0None
1<25%
2>25%
Sub-total Score Substance Factor
TOTAL SCORE (Total of Geometric and Substance Sub-totals)
_______________________________ ____________________
Maximum score = 26 The HIGHER the score, the more severe the ulcer. Evaluator: _________________________________________ Date ___________________________

SCI-PUMT ScoringEach variable assigned ordinal value
Data & clinical judgment to develop cut-points and weights for individual items and total scales score
Determined total score for SCI-PUMT = 26
Assigned proportion of total score to each sub-scale

Surface Area 40%
Depth 14%
Tunneling and Undermining
12% each
Edges, Exudate, Necrotic Tissue
8% each
SCI-PUMT Scoring

Study Limitations
Sample stratification excluded patients who had multiple etiologies of SCI; differentiation of ulcer etiology and ulcer stages were too small for computation
Healing process could be altered by tissue type and ulcer depth
Sample included persons with SCI from one SCI/D Center.

Continuing Psychometric Analysis
Can results of regression model be replicated over time
Does weighting of items improve the SCI-PUMT’s predictive value?
Do subscale scores have clinical utility?

Implications
SCI-PUMT can:◦Help to improve communication
among SCI healthcare providers.◦Form basis for outcomes monitoring
of PrU healing in persons with SCI.◦Assist clinician in critical decisions
affecting overall PrU management.

Implications
◦Allow for comparisons of healing rates within facilities and across sites.
◦Contribute to performance improvement initiatives and local and national performance measures.
◦Provide foundation to conduct treatment effectiveness studies of PrUs in multi-site VA SCI/D Center studies.

Conclusions
This study found that the SCI-PUMT was a reliable, valid, and sensitive
instrument for measuring PrU healing in persons with SCI in a
100 bed VHA SCI/D Center.

The Challenge—Full Implementation of the SCI-PUMT in the VA!