Algae Growth for Biodiesel Fuel Production Jalisa ... · chemical methods, inventions and processes...
Transcript of Algae Growth for Biodiesel Fuel Production Jalisa ... · chemical methods, inventions and processes...

Algae Growth for Biodiesel Fuel Production
Jalisa Richardson
MTH 496: Senior Project
Advisor: Shawn D. Ryan, Asst. Professor, Dept. of Mathematics
Spring 2017

Abstract
As the world and technology continue to grow, there is an increasing need to develop
many different forms of energy. For more than 100 years as a planet the typical form of energy
has been through the burning and consumption of fossil fuels. Fossil fuels are advantageous
because we have the infrastructure in place to continue advancing this technology and it is easy
to cultivate. The disadvantages to the burning and consumption of fossil fuels are the negative
impact on the environment and the fact that this form of energy is not being replaced. With this
in mind there has been a great push to study and implement the use of renewable energy. There
are many different ways to do this including the more known solar, wind, and hydroelectric
power systems. There are also alternative renewable energies such and biomass and biofuel. The
purpose of this project is to investigate and mathematically model the growth of algae in an
attempt to use algae as an alternative source of energy. Algae is an excellent alternative energy
source because it can provide energy in the form of biofuel and it also has the ability to clean
wastewater by consuming minerals during its growth process. This exploration will be done by
developing and analyzing ordinary differential equation based models such as the classic
chemostat model and variations of this model. This paper is organized into several different
sections including an abstract, background information, a brief literature review of current
mathematical models, an extension of these models, results, and a brief discussion/conclusion.

Introduction
Since the turn of the century, rising oil prices have enhanced the need for finding
renewable energy sources that can replace fossil fuel. With that search there has been a
resurfacing of interest in algae biofuels and the increase of funding to create programs on
creating algae biofuel. It was this resurgence on wanting to learn about algae biofuels and the
start of the so-called “green chemistry” movement that helped increase the funding for safe
alternate methods of fuel.
The term “green chemistry” was coined by Paul Anastas in 1991 and can be defined as
“the design of chemical products and processes that are more environmentally friendly and
reduce negative impacts to human health and the environment.”17 Green chemistry takes a look at
chemical methods, inventions and processes that are environmentally friendly and work to either
reduce or eliminate generations of hazardous substances. The concept of green chemistry was
formally established at the Environmental Protection Agency in response to the Pollution
Prevention Act of 1990. Green chemistry can be applied to organic chemistry, inorganic
chemistry, biochemistry, analytical chemistry and even physical chemistry.
There are 12 principles of green chemistry that were developed by Anastas and John C.
Warner, which help explain what green chemistry means in practice.16 The 12 principles are:
• Prevention
• Atom economy
• Less Hazardous Chemical Synthesis
• Designing Safer Chemicals
• Safer Solvent and Auxiliaries
• Design for Energy Efficiency
• Use of renewable feedstock
• Reduce Derivatives
• Catalysis
• Design for Degradation
• Real-time Analysis for Pollution
Prevention
• Inherently Safer Chemistry for
Accident Prevention

The principle of “Prevention” states “it is better to prevent waste than to treat or clean up
waste after it is formed.”16 It is in the best interest of the environment to carry out a synthesis in a
manner that the waste product is minimum or non-existent. Designers need to take into account
the nature of the materials that they are using to ensure that it is either as non-toxic as possible or
uses minimal energy and materials to lessen the environmental and social impacts. The next
principle, “Atom economy”, states, “Synthetic methods should be designed to maximize the
incorporation of all materials used in the process into the final product.”16 In other words atom
economy takes into account the atoms wasted during the chemical processes. The high the atom
economy the more greener the process is deemed to be. Less Hazardous Chemical Synthesis
entails “wherever practicable, synthetic methodologies should be designed to use and generate
substances that possess little or no toxicity to human health and the environment.”16 The goal is
to avoid using known hazardous materials as a starting point, if there are safer alternatives
available. In addition to this, having hazardous waste from these chemical processes is
something that should be avoided, if possible. Designing Safer Chemicals states, “Chemical
products should be designed to preserve efficacy of function while reducing toxicity.”16
Designers must aim to product chemical products that fulfill their function while also having
minimal toxicity to humans. Within this you strive to minimize the toxicity of the materials,
while maintaining its function and efficiency. To reach this goal you must have an understanding
on chemistry, toxicology and environmental science.
Safer Solvents and Auxiliaries states that “The use of auxiliary substances (e.g. solvents,
separation agents, etc.) should be made unnecessary wherever possible and innocuous when
used.”16 While solvents are not always avoidable those that are chosen should reduce the energy

that is required for a reaction, should have minimal or no toxicity and if possible be recyclable
with no major impacts on the environment or safety. Design for Energy Efficiency states that
“Energy requirements should be recognized for their environmental and economic impacts and
should be minimized. Synthetic methods should be conducted at ambient temperature and
pressure.”16 It is better to minimize the energy needed to create a chemical product by having
considerations of the reaction design. By carrying out the reactions at room temperature and
pressure, removal of solvents, or the process used to remove impurities in the products you can
increase the energy that is required for the chemical product and by that increase the process’s
environmental impacts. Use of Renewable Feedstock states that “A raw material or feedstock
should be renewable rather than depleting wherever technically and economically practicable.” 16
Products that last beyond their life cycle are often the cause of environmental issues, such as
petrochemicals and landfills filled with non-recyclable plastics, clothing and other dangerous
materials. By developing and designing products that will withstand their operating purpose
while also being renewable that is non-toxic or dangerous to human life and the environment we
can solve this issue. Reduce Derivatives states that “Reduce derivatives - Unnecessary
derivatization (blocking group, protection/deprotection, and temporary modification) should be
avoided whenever possible”. 16 The best way to reduce derivatives is by using enzymes, since
they often only react with one site of the molecule and leave the rest of it alone. Using this
method protecting groups are not necessarily required. One of the best examples of the use of
enzymes that avoid protecting groups is the synthesis of ampicillin and amoxicillin antibiotics.
Catalysis states, “Catalytic reagents (as selective as possible) are superior to stoichiometric
reagents.” 16 Catalysts do not get used up when they are used by chemical processes, and because
of this they can be recycled many time, and they also do not contribute to waste. Using catalysts

can enable chemical reactions with higher atom economics and because of this they can allow
the utilization of reactions that wouldn’t be possible under normal circumstances, but also
produce less waste. Design for Degradation states that “Chemical products should be designed so
that at the end of their function they do not persist in the environment and break down into
innocuous degradation products.” 16 This is an ideal principle because when you have products
that do not break down once they have fulfilled their use they typically accumulate and remain in
the environment. Chemical products should be designed in a way that allows them to break down
into harmless products so they will not contribute to the harm of the environment. ” Real-time
Analysis for Pollution Prevention states that “Analytical methodologies need to be further
developed to allow for real- time, in-process monitoring and control prior to the formation of
hazardous substances.” 16 By monitoring the chemicals when they have a reason we are able to
help prevent the release of hazardous substances that arise from accidents or unexpected
reactions. With this, warning signs can be spotted, changes can be detected and other events can
be prevented before a major incident even occurs. Inherently Safer Chemistry for Accident
Prevention states that “Substances and the form of a substance used in a chemical process should
be chosen to minimize potential for chemical accidents, including releases, explosions, and fires.”
16 When you work with chemicals there is always a level of risk attached to it, but if you can
manage those chemicals and hazards well, the risk can be minimalized for those involved. This
principle links a number of other principles that discuss hazardous materials. If it is possible
exposure to hazardous materials should be removed from processes, and if removal is not
possible should be designed to minimize the risk associated with them.

Algae as Biofuel
In 1942 it was proposed that algae could be used as a source of lipids for food or fuel.
Research began in the U.S., Germany, Japan, England and Israel following World War II on
discovering techniques, designing and engineering systems for growing algae large scale. After
time passed from WWII there was a period of time that there was no need for research on algae
as a form of biofuel but rather utilizing algae as a source of food or method for water treatment.
Interest in algae as biofuels resurfaced during the 70’s energy crisis, leading to the US
Department of Energy to create the Aquatic Species Program (ASP) in 1978. Over the years ASP
arrived to prove that microalgae would be more effective and efficient in the world, but there
were issues with the program. While the cultivating of algae was attainable, outdoor ponds were
venerable to environmental disturbances. The next instance is when they realized that that the
cost of unextracted algae oil would not be competitive with petroleum oil. After trying for years
the ASP was abandoned until a solution in making algae competitive was found.
It wasn’t until the 2000s that rising oil prices resurfaced the interest in algae biofuels and the
increase of funding to create programs on creating algae biofuel. It was this resurgence on
wanting to learn about algae biofuels and the start of the “Green Chemistry” movement that
helped increase the funding for safe alternate methods of fuel. Algae biofuel is still a relatively
new technology and the disadvantage in that is all research utilizing it as fuel has been on a
small-scale measure that is no sufficient to support our current energy needs.
The process of creating biofuel from algae is a long and extensive process with many
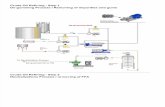
different aspects that must be considered. In the figure below, we can see how many different
steps this process must go through before it is considered biofuel. This figure does not include
the packaging and distribution of the final product.

Figure 1: Process flow diagram production of biodiesel from algae.3
From the figure above there are many different piece of equipment used during the entire
process. This equipment contributes to a sizable initial monetary investment, which is why many
companies are not able to make the switch to produce algae for biofuel/biodiesel purposes. But,
many companies are now seeing the need and benefit for the production of algae are employing
many different ways to produce, cultivate, and harvest the algae is the most economically and
environmentally safe way possible. Society today has an apparent dependence on fossil fuels for
means of energy, transportation, and industrial production. However, the high usage of non-
renewable fossil fuels has caused concerns not only due to limited supply, but the harmful
emissions burning fossil fuels can cause. Biomass has become the newest alternative to fossil
fuels, and its conversions to bioethanol and biodiesel are now often being implemented in many
industries. That being said, many options are available in terms of which types of biomass would

be the best selection. Depending on the biomass used, issues such as transportation, “food vs.
fuel” allotment, and soil degradation become major concerns in whether or not biofuels have a
lasting chance as adequate replacement for fossil fuels.
Cultivation of Algae
For our process we will be using microalgae to transform radiant energy into products
that we can use for everyday resources. The need for the development of this process came from
two main driving forces. The first is that there has been a need for alternative resources of
several products, due to political or economic reasons4. Algae are considered one of the most
advantageous renewable resources because of its use of relatively inexpensive resources such as
CO2 and H2O. Other major advantages are the potential for reduction of emissions from power
plants, much higher productivity than traditional fast-growing energy crops, less area required
for growth when grown in a photo-bioreactor, the use of photo-bioreactor prevents potential
degradation of soil and groundwater, and wastewater can be used which aids in wastewater
treatment5.
Table 1: Main design features of open and closed systems. These features are important to consider when
optimizing the output of algae from either open ponds or bioreactors4.
The cultivation of algae has been primarily done using open ponds. They can either be
configured as circular ponds with a rotating arm to mix the culture, or long channels in a single
or multiple loop configurations stirred by a paddle wheel4. Open ponds are difficult to work with

because it is almost impossible to control contamination, it is difficult to keep the environment
constant, and it is expensive to harvest the crop. The weather is also an unpredictable factor that
can greatly affect the yield of algae produced. With these constraints in mind, another approach
has been developed which is based on using a closed system. Closed systems are beneficial
because they allow sensitive strains of algae to be grown, the control of contaminants is more
easily manageable, and the cost of cultivation of the product is greatly reduced because of the
higher cell mass productivities attained4. There are also some disadvantages to using a closed
system for the growth and harvesting of algae. Primarily, the capital cost for the equipment
needed is substantially higher than that of an open system. There is also the increased production
cost because there is an added cost for the supply of light and CO2 to operate the photo-
bioreactor. For this process, nutrient supply, pH, temperature control, and harvesting are also
important aspects that could be included in the cost of operation.
Mathematical Approach
For the purpose of this project, an ordinary differential equation (ODE) model will be
employed in order to model the optimization of the algae growth. The actual growth of algae is
simple, and includes carbon dioxide being pumped into water and then converted into energy or
sugar by way of photosynthesis6. Nutrients that are in the wastewater are then combined with the
energy or sugars to form more algae, thus an ever-growing system is created6.
Figure 2: This is a simple representation of the growth of algae from nutrients and carbon dioxide
The above figure includes many of the different parameters that are considered during the
algae growth process. The concentration of dry algae (A), nutrients (M), sugar (S), and carbon

dioxide (C) are all-dependent on time t6. Im is the flow rate of the nutrients and Ic is the flow rate
of the carbon dioxide. The process also considers Dr and hr, which are the death rate and harvest
rate respectively6. These parameters are also important because they model the reduction of algae.
The sugar or energy is modeled using the rate of αsC where αs is the constant rate and the amount
of carbon dioxide is decreased by a rate of –k1αsC. There is also the production of new algae
from already existing algae by a rate of αANfm(M) from nutrients and sugar where αA is another
constant rate and fm(M) denotes the concentration of nutrients inside the cells6. In order to create
the system of ODEs used the algae growth, photosynthesis, flow rate of carbon dioxide and
nutrients, and the removal of algae was combined6.
Table 2: The system of ODEs used to model growth of algae and its parameters6.
Similarities in Models
The chemostat model is a simpler way to describe the growth of algae with a given concentration
of algae and concentration of nutrients. In a chemostat model the growth of the algae is based on

the limiting nutrients alone. The ODE model above and the chemostat model depend on various
components. The chemostat model uses V for the volume of the chemostat, Q is the volumetric
flow rate, 𝑞 = 𝑄
𝑉 is the dilution rate, N(t) is the mass of bacteria, c(t) is the concentration of the
rate limiting nutrient, and r is the growth rate. With these parameters we can decrease the set of 4
differential equations into just a coupled set of ordinary differential equations to model the algae
growth process in the pond environment.
𝑑𝑁(𝑡)
𝑑𝑡= 𝑟(𝑐(𝑡))𝑁(𝑡) − 𝑞𝑁(𝑡) (1)
𝑑𝑐(𝑡)
𝑑𝑡= 𝑞𝑐0 − 𝑞𝑐(𝑡) −
1
𝑦𝑟(𝑐(𝑡))𝑁(𝑡) (2)
We also introduce the term 𝑞𝑐0 which is the input rate of the initial concentration and y as the
yield parameter. The reaction kinetics are also an important factor to be considered and can be
determined using the Michaelis-Menten reaction kinetics where
𝑟(𝑐) = 𝑅𝑚𝑎𝑥𝑐
𝐾𝑚 + 𝑐
In order to find the steady state conditions used to operate the chemostat model equations (1) and
(2) are manipulated to get:
0 = 𝑟(𝑐̅(𝑡))𝑁(𝑡) − 𝑞𝑁(𝑡) (3)
0 = 𝑞𝑐0 − 𝑞𝑐̅(𝑡) −1
𝑦𝑟(𝑐̅(𝑡))�̅�(𝑡). (4)
The steady state can be found once the constants are found that allow the below mathematical
equations to be true
𝑑𝑁
𝑑𝑡|
(�̅�,𝑐̅)= 0,
𝑑𝑐
𝑑𝑡|
(�̅�,𝑐̅)= 0.

First solve 𝑟(𝑐̅)𝑁 − 𝑞𝑁 = 0 from here we can assume that 𝑁 is non-zero so 𝑟(𝑐̅) = 𝑞. Next we
can substitute r(𝑐̅) to obtain
𝑞 = 𝑅𝑚𝑎𝑥𝑐̅
𝐾𝑚 + 𝑐̅
Where ultimately we can obtain the relationship 𝑐̅ = 𝐾𝑚𝑞
𝑅𝑚𝑎𝑥−𝑞
Next, we solve 0 = 𝑞𝑐0 − 𝑞𝑐̅(𝑡) −1
𝑦𝑟(𝑐̅(𝑡))�̅� (𝑡) since 𝑟(𝑐̅) = 𝑞. Through basic algebra we
can see that 0 = 𝑐0 − 𝑐̅ −1
𝑦𝑁 and 𝑁 = 𝑦(𝑐0 − 𝑐̅). This would give the nontrivial steady state
(𝑁, 𝑐̅) = (𝑦(𝑐0 − 𝑐̅),𝐾𝑚𝑞
𝑅𝑚𝑎𝑥−𝑞) (5)
It is important to note that there is also a trivial steady state in which (N,c) = (0, c0). This
particular steady state is disregarded because it would imply that N = 0 and creates an unstable
equilibrium. It is unstable because c depends on c0 which means that there will be a different
equilibrium as the c0 changes. Also since y ≠ 0 (0, c0) will never converge and since we are
forced to start at a point that is not the equilibrium c0 ≠ c so N ≠ 0. This implies that (0, c0) is an
unstable equilibrium while the nontrivial state is a stable equilibrium.
Graphical Results
In order to graphically support what was depicted analytically, the ODE equations from the
original four-equation system were put into MATLAB. The results were also compared to the
logistic model for the growth of algae.

Figure 4: The results of the numerical model(Black) and the logistic equation (red)6.
Table 3: Guessed and typical values based on past literature
Initial values were also chosen at A(0) = 6 g[A] m-3 , M(0) = 4g[M]m-3, C(0) = 7g[CO2 ] m-3 and
S(0) = 10g[S]m-3 6. These values chosen correctly modeled the optimization of the growth of
algae. This shows that algae would be best grown in a two-pond system that will allow for the
continuous cleaning of the wastewater created when the nutrients are recycled and absorbed
during the process.
Extension of Work
The extension of the above process is to design a pond system for the growth and harvesting of
algae. Then, model the growth using established parameters, Matlab and the simplified
chemostat model. Some of the parameters will be identical as they are ideal parameters for the
growth of algae.
For the process there will be a flue gas used with content of CO2 of 12.5 vol%. This is the
maximum value for a gas-fired power station14. The flue gas would be the source of the carbon.

The goal of the process is the production of microalgae with high contents of
Triacylglycerides(TAG). In order to achieve this goal a two-stage approach was implemented
where in the first stage the culture would be grown under nutrient-sufficient circumstances in
order to obtain a highly density culture, then the culture would be transferred to another
raceway/pond where the nitrogen source is not available14. The culture would then be left until
the TAG content is increased to the desired concentration level. Once that level is obtained the
crop would be harvested and sent to the next step, the flocculation step. Both raceways would
operate continuously (8hr/day) while the photosynthesis process occurs and then held as a batch
reaction for the remaining hours of the day. In order to determine the nutrient requirements for
the stage 1 raceway of the process, the algal composition of CH1.83O0.48N.11P.01 was calculated14.
Nitrogen would be provided in the form of ammonium nitrate, which contains 34.5 wt.%
nitrogen, and the fertilizer calcium dihydrogen phosphate also known as triple super phosphate
fertilizer will be used to provide the phosphorous. This fertilizer contains 18 wt.%
phosphorous14. The raceway would be built above ground using concrete. The external walls and
a central divider of the raceways would then have a lifetime of 20 years. The density of the
concrete hollow blocks would be 650 kg/m3 and have dimensions of .44 x .22 x .215 14. Also, a
white PVC pipe of 0.75 x 10-3m thickness and lifetime of 5 years would be installed14.
This PVC pipe would be installed to enhance uniform flow rates and to avoid re-
suspension of sediments that have settled on the bottom of the raceway to keep the water as
clean/clear as possible. Also, a paddle wheel would be installed as typically used to provide the
essential mixing. The paddle wheel would provide a typical electrical efficiency of 17%14. The
electrical efficiency is the useful power output/total power input. The assumed design parameters
for raceway 1 are as follows; depth: 0.3m, length: 150m, width:10m, hydraulic mean depth:

.28m, pond area: .33 ha, pond volume 1000 m3, mean liquid velocity: 3 m/s, Reynolds number
85000, flue gas(STP): 1445 m3/day, outlet flow rate: 100 m3/day and outlet biomass density: 1
kg/m3. The assumed design parameters for raceway 2 are as follows; depth: 0.3m, length: 190 m,
width: 20 m, hydraulic mean depth: .28 m, pond area: .88 ha, pond volume 2700 m3, mean liquid
velocity: 03 m/s, Reynolds number 87000, flue gas(STP): 1926 m3/day, outlet flow rate: 200
m3/day and outlet biomass density: 1.7 kg/m3. Based on these assumed parameters a final
concentration of TAG was calculated to be 40 wt.% of the dry cell, this equates to 40 tons ha-1
year-1(1 ha = 104 m2)4.
Mathematical Modeling
As mentioned above, the modeling of the growth of algae will be done using the Chemostat
model.
𝑑𝑁
𝑑𝑡= 𝛼1
𝐶
1+𝐶𝑁 − 𝑁 (6)
𝑑𝐶
𝑑𝑡= −
𝐶
1+𝐶𝑁 − 𝐶 + 𝛼2 (7)
Using this model N and C are dimensionless and represent bacteria and nutrients. Also 𝛼1 =
𝐾𝑚𝑎𝑥𝑉
𝐹 and 𝛼2 =
𝐶0
𝐾𝑛 Where Kmax and Kn are based on the Michaelis-Menten kinetics.
Parameter Set
1
Parameter Set 2
Kmax 5.8 Kmax 5.8
Kn 2.9 Kn 2.9
V 1000m3 V 2700m3
F 1445m3/day F 1926m3/day
C(0) 10 C(0) 2
N(0) 1 N(0) 20
Alpha1 4.01 Alpha1 1926
Alpha 2 .344 Alpha 2 .689 Table 4: Calculated and researched values
Results
We proceed to use Matlab to simulate the above two equation Chemostat model with parameter
sets 1 ans 2 given in Table 4. See figures for additional detail.

Figure 5: MATLAB picture depicting the growth of algae in pond 1
Figure 6: MATLAB picture depicting the growth of algae in pond 2
Based on the chemostat model of the selected parameters, the pond 1 would see N(t) die off as
time progressed. In order to change that either the volume would increase or increase the initial
amount of bacteria. In pond 2 we can see that the N(t) reached a limit of 1400, it is difficult to

see how exactly the concentration is modeled because it such a smaller number than the N(t)
growth, but for the purpose of this model, we want the nutrients to grow substantially and to
maintain a stable level of growth. As depicted in the above graphs the algae would first be grown
in pond 1 once it reaches its maximum concentration the algae would then be harvested and
transferred to pond 2 where the concentration of algae will be sustained in pond 2.
Other Initial Conditions
α1 and α
2 are important aspects of the chemostat model as it pertains to how the model is
manipulated in MATLAB. As stated before we have two steady state conditions, one is trivial
while the other in non-trivial. We disregard the trivial steady state because it states that there is
no bacterial left and the concentration remains the same as the initial concentration. With that in
mind we manipulate the non-trivial steady state solution but there are still biological aspects to
consider. In particular, α1
> 1, this is true because biologically you can never have a negative
population or concentration. Since α1
> 1 then α2
>1
(α1
−1) must also be true. With this in
mind it is important to explore other initial conditions to determine the limits of the chemostat
model. First, with the below parameters in mind
Figure 7: MATLAB picture depicting the growth of algae when all conditions are met
C(0) .6
N(0) .01
Alpha1 2.5
Alpha 2 2

It can be seen that from satisfying these 4 simplified parameters, the Chemostat model correctly
depicts the growth on algae in a chemostat chamber. Next, with the below parameters in mind,
Figure 8: MATLAB picture depicting the growth of algae when 𝜶𝟏 is not greater than 1
It is important to note that because α1 is not greater than 1 the concentration of bacteria
continues to grow until it reaches a steady state while the N(t) almost immediately dies off which
is not ideal for the modeling of the growth of algae. Finally, with the below parameters in mind
Figure 9: MATLAB picture depicting the growth of algae when the conditions for 𝛂𝟐 is not met
C(0) .6
N(0) .01
Alpha1 .5
Alpha 2 1.0
C(0) .6
N(0) .1
Alpha1 2.0
Alpha 2 .8

Since 𝛼2 >1
(𝛼1−1) is not satisfied in this case then again the N(t) immediately dies off instead of
growing, which is not ideal for the modeling of the grown of algae in a chemostat chamber.
Conclusion
After researching available biomass options, it was determined that algae were the best
candidates to be used in the production of biofuel. Algae was selected for its high yields and low
carbon footprint. A two-raceway model was selected because it has been found to produce the
most algae. As mentioned before algae are made when CO2 converted to energy and then
minerals and that energy is combined to create more algae. The parameters for the chemostat
model were chosen based on research. The benefits of using the chemostat model is that it is
simpler than other systems of ODE and that the parameters can be adjusted as needed based on
the size of the pond, flow rate and the initial values for the bacteria and nutrients. For an
extension on this project, there is a possibility to model the exact amount of algae that can be
grown to replace 20% of the energy used by Cuyahoga County in Ohio.

References
1. Tredici, M.R. & Materassi, R., “From open ponds to vertical alveolar panels: the
Italian experience in the development of reactors for the mass cultivation of phototrophic
microorganisms,” Journal of Applied Phycology 4: pp. 221-231(1992).
2. Pushparaj, Benjamin, et. al., “An integrated culture system for outdoor production of
microalgae and cyanobacteria,”Journal of Applied Phycology 9: pp.113-119(1997).
3. Stephenson, Anna, et. al., “Life-Cycle Assessment of Potential Algal Biodiesel
Production in the United Kingdom:A Comparison of Raceways and Air-Lift Tubular
Bioreactors,” Energy Fuels 24: pp. 4062-4077(2010).
4. Carvalho, Ana P, et. al., “Microalgal Reactors: A Review of Enclosed System Designs
and Performances,” Biotechnol. Prog. 22: pp. 1490-1506(2006).
5. Cuello, Joel L., Mason, Michael, and Kazz, Michael., “Design of scalable
photobioreactors for Mass production of Algae for Biofuel Production,”University of
Arizona. (2006).
6. Thornton, Anthony., Weinhart, Thomas, & Bokhove, Onno. “Modeling and
optimization of algae growth” Universiteit Twente. (2011).
7. United States Department of Energy. Web.
http://www1.eere.energy.gov/biomass/pdfs/algalbiofuels.pdf.
8. "Celsias." Particles in the Pool, Fuel in the Tank: The Layman's Guide To Algae
Biofuel. N.p.. Web.http://www.celsias.com/article/particles-pool-fuel-tank-laymans-
guide-algae-biofu/.
9. Ciampa, Frank. "Algae Biodiesel: Pros and Cons." The Green Economy Post. N.p., n.d.
Web.http://greeneconomypost.com/algal-biodiesel-pros-and-cons-9573.htm.

10. Holle, Sieg. "Skimaire High Tech." Algae waste water treatment to green energy ideas.
N.p., 19 2010. Web.http://skimairecanada.blogspot.com/2010/12/algae-waste-water-
treatment-to-green.html.
11. Hossain, Sharif. "American Journal of Biochemistry and Biotechnology." American
Journal of Biochemistry and Biotechnology . 4. (2008): 254.
Web.http://www.aseanbiotechnology.info/Abstract/21029966.pdf .
12. Newman, Stefani. "How Algae Biodiesel Works." 18 June 2008.
HowStuffWorks.com.http://science.howstuffworks.com/environmental/green-
science/algae-biodiesel.htm.
13. Patil, Vishwanath. "International Journal of Molecular Sciences." International Journal
of Molecular Sciences. 9.7 (2008): 1188-1195. Web.http://www.mdpi.com/1422-
0067/9/7/1188/htm
14. Rhodes, Chris. "Why Algal Biofuels May Never Hold the Key to the Future."
OilPrice.com. CNBC, 19 2012. Web.http://oilprice.com/Alternative-
Energy/Biofuels/Why-Algal-Biofuels-May-Never-Hold-the-Key-to-the-Future.html .
15. University of Virginia. “Engineers find significant environmental impacts with algae-
based biofuel.” ScienceDaily, 25 Jan. 2010.
Web.http://www.sciencedaily.com/releases/2010/01/100121135856.htm.
16. 12 Principles of Green Chemistry. (n.d.). Web.
https://www.acs.org/content/acs/en/greenchemistry/what-is-green-
chemistry/principles/12-principles-of-green-chemistry.html
17. Green Chemistry. (2016, December 19). Web. https://www.epa.gov/greenchemistry