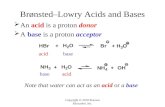AcidBase
-
Upload
doxa-dave-rotap -
Category
Documents
-
view
241 -
download
3
Transcript of AcidBase

1
Acid-Base PropertiesOBJECTIVES
of
Organic Compounds
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
EXPERIMENT
5
ARMSALCEDO [email protected]
IR ANALYSIS
EXPERIMENT
5
ARMSALCEDO [email protected]
Objectives
to describe the structural features common to compounds that belong to a specific functional grouping
to be able to differentiate each component of a given mixture of organic functionality based on solubility and acid-base properties
OBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
IR ANALYSIS

2
EXPERIMENT
5
ARMSALCEDO [email protected]
Highlighted Concepts
can be determined without changing the chemical identity of a
substance
examples are physical color, odor, physical state, solubility
and density
Physical PropertiesPhysical PropertiesOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
IR ANALYSIS
EXPERIMENT
5
ARMSALCEDO [email protected]
Highlighted Concepts
Acids and BasesAcids and Bases
Brönsted-LowryBrönsted-Lowry LewisLewis
AcidsAcids
BasesBases
Proton donor
Proton acceptor
Electron pair acceptor
Electron pair donor
OBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
IR ANALYSIS

3
EXPERIMENT
5
ARMSALCEDO [email protected]
Highlighted Concepts
Acid-Base propertiesAcid-Base propertiesOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
AcidsAcids BasesBasessour in taste
increase H+ concentration
taste bitter and feel soapy
increase OH- concentration
“Organic acids and bases are weak in nature and don’t
ionize completely in H2O.”IR ANALYSIS
EXPERIMENT
5
ARMSALCEDO [email protected]
Highlighted Concepts
Acid-Base properties of organic compoundsAcid-Base properties of organic compoundsOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
organic acids and bases are weak in nature and don’t ionize
completely in H2O
organic compound’s acid – base behavior can be obtained by
solubility comparison in aqueous acids and aqueous bases
IR ANALYSIS

4
EXPERIMENT
5
ARMSALCEDO [email protected]
Highlighted Concepts
SolubilitySolubilityOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
ability of a substance to dissolve
solute – the substance being dissolve
solvent – a substance in which other substances are
dissolved
solution – mixture of solute and solvent
maximum amount of solute that dissolves in a solvent at
equilibrium
IR ANALYSIS
EXPERIMENT
5
ARMSALCEDO [email protected]
Highlighted Concepts
Factors affecting solubilityFactors affecting solubilityOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
TEMPERATURE
around 95% of solid solutes, the solubility increases with
temperature
PRESSURE
the solubility of a gas in a liquid is directly proportional to
the partial pressure of that gas above the liquid (Henry’s
Law)
POLARITY
“Like dissolves like.”
IR ANALYSIS

5
EXPERIMENT
5
ARMSALCEDO [email protected]
Highlighted Concepts
Solubility vs MiscibilitySolubility vs MiscibilityOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
SOLUBILITY - a measure of one substance's ability to
dissolve in a specific amount of another substance at standard
temperature and pressure
MISCIBILITY - a measure of how easily different liquids will
dissolve when mixed together
IR ANALYSIS
EXPERIMENT
5
ARMSALCEDO [email protected]
ReagentsOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
Amyl acetate
Cyclohexane
EthylamineEthanol
Benzoic acidPhenol
IR ANALYSIS
Reference StandardReference Standard

6
EXPERIMENT
5
ARMSALCEDO [email protected]
ReagentsOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
Hydrochloric acid (HCl)
strong acid because it can be completely dissociated with
water
Sodium hydroxide (NaOH)
strong metallic base which reacts with strong acids to form
water and its corresponding salts
Sodium bicarbonate (NaHCO3)
white crystalline solid which when treated with acids
decomposes to water and CO2
IR ANALYSIS
EXPERIMENT
5
ARMSALCEDO [email protected]
ProcedureOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
Amyl acetate
Cyclohexane
Ethylamine
Ethanol
Benzoic acid
Phenol
5 drops or
microspatula
physical state
color
odor
Physical PropertiesPhysical Properties
IR ANALYSIS

7
EXPERIMENT
5
ARMSALCEDO [email protected]
ProcedureOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
1. Amyl acetate
2. Cyclhexane
3. Ethylamine
4. Ethanol
5. Benzoic acid
6. Phenol
Solubility/Miscibility TestSolubility/Miscibility Test
+ 1 mL 1M HCl
+ 1 mL 1M NaOH
Observe
and record
the
solubility of
each
compound
1 2 3 4 5 6
1 2 3 4 5 6
IR ANALYSIS
EXPERIMENT
5
ARMSALCEDO [email protected]
ProcedureOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
Benzoic acid Phenol
Solubility/Miscibility TestSolubility/Miscibility Test
Observe
and record
the
solubility of
each
compound
NaHCO3
+ 10
drops
microspatula 10 drops
Observe for any effervescence.IR ANALYSIS

8
EXPERIMENT
5
ARMSALCEDO [email protected]
ResultsOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
Organic Sample
Physical Properties Miscibility
Physical State
Odor, ColorWith 1M HCl*
With 1M NaOH*
_ _ _Amyl acetate Liquid
Before heating:
After heating:
immiscible immiscibleBanana-like odor; Colorless.
Banana-like odor;
Colorless.
Cyclohexane LiquidGasoline-like odor ;
Colorless immiscible immiscible
Ethylamine LiquidAmmonia-like odor;
Colorless miscible miscible
Ethanol LiquidAlcohol-like odor;
Colorless miscible miscible
With NaHCO3*
Benzoic Acid SolidOdorless; White, fine
powder insoluble soluble
-With
effervescence
Phenol LiquidFormalin-like odor; Light
red immiscible miscible
-Without
effervescence
IR ANALYSIS
EXPERIMENT
5
ARMSALCEDO [email protected]
DiscussionOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
Amyl AcetateAmyl Acetate
neutral compound
participate in H-bond as hydrogen-bond acceptors, but cannot
act as hydrogen-bond donors
ester molecules cannot hydrogen-bond to each other
immiscible in both HCl and NaOH
IR ANALYSIS

9
EXPERIMENT
5
ARMSALCEDO [email protected]
DiscussionOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
CyclohexaneCyclohexane
neutral compound
cannot form H bonds because of their non-polarity
immiscible in both HCl and NaOH
IR ANALYSIS
EXPERIMENT
5
ARMSALCEDO [email protected]
DiscussionOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
EthanolEthanol
can form H-bond
considered neutral compound but may act as acid or base
can dissolve in both polar and non-polar substances
miscible with HCl and NaOH
IR ANALYSIS

10
EXPERIMENT
5
ARMSALCEDO [email protected]
DiscussionOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
EthylamineEthylamine
can form H-bond
act as a base
basicity depends on the availability of the lone pair of electrons
of N and on the electronic properties of the substituents
miscible with HCl and NaOH
Reaction of Ethylamine with HCl
CH3CH2NH2 + HCl � CH3CH2NH3+ + Cl-
IR ANALYSIS
EXPERIMENT
5
ARMSALCEDO [email protected]
DiscussionOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
Benzoic acidBenzoic acid
can form H-bond
act as an acid
soluble in NaOH
insoluble in HCl and forms effervescence with NaHCO3
Reaction of Benzoic acid with NaOH
C6H5COOH + NaOH � C6H5COO-Na+ + H2OIR ANALYSIS

11
EXPERIMENT
5
ARMSALCEDO [email protected]
DiscussionOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
PhenolPhenol
can form H-bond
slightly acidic
miscible in NaOH
immiscible in HCl and does not form effervescence with
NaHCO3
Reaction of Phenol with NaOH
IR ANALYSIS
EXPERIMENT
5
ARMSALCEDO [email protected]
Infrared AnalysisOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
EthylacetateEthylacetate
IR ANALYSIS

12
EXPERIMENT
5
ARMSALCEDO [email protected]
Infrared AnalysisOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
CyclohexaneCyclohexane
IR ANALYSIS
EXPERIMENT
5
ARMSALCEDO [email protected]
Infrared AnalysisOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
EthylamineEthylamine
IR ANALYSIS

13
EXPERIMENT
5
ARMSALCEDO [email protected]
Infrared AnalysisOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
EthanolEthanol
IR ANALYSIS
EXPERIMENT
5
ARMSALCEDO [email protected]
Infrared AnalysisOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
Benzoic acidBenzoic acid
IR ANALYSIS

14
EXPERIMENT
5
ARMSALCEDO [email protected]
Infrared AnalysisOBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
PhenolPhenol
IR ANALYSIS
EXPERIMENT
5
ARMSALCEDO [email protected]
OBJECTIVES
REAGENTS
PROCEDURE
RESULTS
CONCEPTS
DISCUSSION
IR ANALYSIS
PREPARED BY:PREPARED BY:PREPARED BY:PREPARED BY: