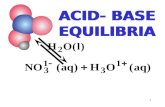Acid base imbalance in medicine
-
Upload
omar-danfour -
Category
Health & Medicine
-
view
4.695 -
download
4
Transcript of Acid base imbalance in medicine

1
Acid- Base Balance
Dr. Omar Mohamed DanfourSenior Lecturer –Specialist of Anesthesia & Intensive Care
(MSU-IMS-Anesthesia Department

• Acids are H+ donors.• Bases are H+ acceptors, or give up OH- in solution.• Acids and bases can be:
– Strong – dissociate completely in solution • HCl, NaOH
– Weak – dissociate only partially in solution• Lactic acid, carbonic acid 2
• PH = - log [H+]• H+ is really a proton• If [H+] is high, the solution is acidic pH
• If [H+] is low, the solution is basic or alkaline pH
pH Review

Acid Base -Basic Concepts• Hydrogen Ion [H+] is tightly controlled • [H+] is determined by the balance between PaCO2
and serum HCO3 (bicarbonate), {normal ratio is 20 (Hco3) : 1(H2Co3)}
Henderson-Hasselbalch Equation [H+] = 24 (PaCO2 / HCO3
-)Normal Values
– [H+] = 40 nEq/L – pH = 7.40 (7.35-7.45)– PaCO2 = 40 mm Hg (35-45)– HCO3 = 24 mEq/L (22-26)– < 6.8 or > 8.0 death occurs 3

[H+] pH
pH = 6.1 + log ([PaCO2] / [0.03 x HCO3-])
pH [H+] pH [H+]
7.807.75
1618
7.307.25
5056
7.707.65
2022
7.207.15
6371
7.607.55
2528
7.107.00
7989
7.507.45
3235
6.956.90
100112
7.407.35
4045
6.856.80
141159 4

Keep It Simple• PaCO2 = Acid
– PaCO2 = pH (Acidemia)– PaCO2 = pH (Alkalemia)
• HCO3 = Base– HCO3 = pH (Alkalemia)– HCO3 = pH (Acidemia)
• Acidosis: pH < 7.35– Respiratory PaCO > 40 mmHg– Metabolic HCO3 < 24 mEq/L
• Alkalosis: pH > 7.45– Respiratory PaCO2 < 40 mmHg– Metabolic HCO3 >24 mEq/L
5

6
The Body and pH

Simple Acid-Base Disorders
Type of Disorder pH PaCO2 [HCO3]
Metabolic Acidosis Metabolic Alkalosis Acute Respiratory Acidosis Chronic Respiratory Acidosis Acute Respiratory Alkalosis Chronic Respiratory Alkalosis
7

Small changes in pH can produce major disturbances
• Most enzymes function only with narrow pH ranges• Acid-base balance can also affect electrolytes (Na+,
K+, Ca++, Cl-)• Can also affect hormones• The body produces more acids than bases• Acids take in with foods• Acids produced by metabolism of lipids and
proteins• Cellular metabolism produces CO2.
• CO2 + H20 ↔ H2CO3 ↔ H+ + HCO3-
8

Control of Acids1. Buffer systems
Take up H+ or release H+ as conditions change Buffer pairs – weak acid and a base Exchange a strong acid or base for a weak one Results in a much smaller pH change
CO2 + H20 ↔ H2CO3 ↔ H+ + HCO3-
Bicarbonate buffer• Sodium Bicarbonate (NaHCO3) and carbonic acid (H2CO3)
• Maintain a 20:1 ratio : HCO3- : H2CO3
HCl strong Acid + NaHCO3 weak Base ↔ H2CO3 weak acid + NaCl
NaOH Sttrong base + H2CO3 weak acid ↔ NaHCO3 weake base + H2O 9

Phosphate buffer• Major intracellular buffer• H+(acid) + HPO4
2- ↔ H2PO4- (titratable acids eliminated in urine)
• OH-(Base) + H2PO4-acid ↔ H2O + H2PO4
2-
Protein Buffers• Hemoglobin is rich in histidine which is an effective buffer from
PH5.7 to 7.7.(Hb in RBCs in equilibrium as a weak acid(HHB) and a potassium salt (KHb)
• Carboxyl group gives up H+ • Amino Group accepts H+
• Side chains that can buffer H+ are present on 27 amino acids.
10

2. Respiratory mechanisms• Exhalation of carbon dioxide• Powerful, but only works with volatile acids• Doesn’t affect fixed acids like lactic acid• CO2 + H20 ↔ H2CO3 ↔ H+ + HCO3
-
• Body pH can be adjusted by changing rate and depth of breathing
3. Kidney excretion• Can eliminate large amounts of acid• Can also excrete base• Can conserve and produce bicarb ions• Most effective regulator of pH• If kidneys fail, pH balance fails
11

12
• Buffers function almost instantaneously (rapid)
• Respiratory mechanisms take several minutes to hours
• Renal mechanisms may take several hours to days
Rates of correction

13

Acid-Base Imbalances• pH< 7.35 acidosis• pH > 7.45 alkalosis• The body response to acid-base imbalance is called
compensation• May be complete if brought back within normal
limits• Partial compensation if range is still outside normals• If underlying problem is metabolic, hyperventilation
or hypoventilation can help : respiratory compensation.
• If problem is respiratory, renal mechanisms can bring about metabolic compensation 14

15

Respiratory Acidosis• Carbonic acid excess caused by blood levels of CO2
above 45 mm Hg. • Hypercapnia – high levels of CO2 in blood
Causes:• Chronic conditions:
– Depression of respiratory center in brain that controls breathing rate – drugs or head trauma
– Paralysis of respiratory or chest muscles– COPD, pneumonia & obesity
16
CO2 + H20 ↔ H2CO3 ↔ H+ + HCO3-

Respiratory Acidosis
• Acute conditons:– Adult Respiratory Distress Syndrome– Pulmonary edema– Pneumothorax– Pulmonary emboli– Aspiration pneumonia– Increased CO2 production (Malignant
hyperthermia & thyroid storm)17

Signs and Symptoms of Respiratory Acidosis
• Breathlessness• Restlessness• Lethargy and disorientation• Tremors, convulsions, coma• Respiratory rate rapid, then gradually
depressed• Skin warm and flushed due to vasodilation
caused by excess CO218

Compensation for Respiratory Acidosis
• This is accomplished via two mechanisms; a) rapid cell buffering and• In this setting, carbonic acid (H2CO3 ) can only be
buffered by the limited intracellular buffers (primarily hemoglobin and proteins). H2CO3 + Hb- → HHb + HCO3-
b) an increase in net acid excretion.• Kidneys eliminate hydrogen ion and retain
bicarbonate ion (Chronic state) 19

20

Treatment of Respiratory Acidosis
• Restore & improve alveolar ventilation• IV lactate solution (converted to bicarbonate
ions in the liver). • Treat underlying dysfunction or disease e.g. pul odema, Res depression
21

Respiratory Alkalosis
• Carbonic acid deficit• pCO2 less than 35 mm Hg (hypocapnea)
• Most common acid-base imbalance• Primary cause is hyperventilation
22
CO2 + H20 ↔ H2CO3 ↔ H+ + HCO3-

Respiratory Alkalosis• Conditions that stimulate respiratory center:
– Oxygen deficiency at high altitudes– Pulmonary disease and Congestive heart failure – caused
by hypoxia – Acute anxiety & pain– Fever, anemia– Early salicylate intoxication– Cirrhosis– Gram-negative sepsis– Iatrogenic (ventilator induced)
23

• Alkalosis causes over excitability of the central and peripheral nervous systems.
• Numbness• Light headedness• It can cause :
– Nervousness– muscle spasms or tetany – Convulsions – Loss of consciousness– Death
24
Signs and Symptoms of Respiratory Alkalosis

Compensation of Respiratory Alkalosis• There are two mechanisms responsible for this
compensation to respiratory alkalosis; 1) Rapid cell buffering and 2) Decrease in net renal acid excretion.• hydrogen ions move from the cells into the extracellular
fluid, where they combine with [HCO3- to form carbonic acid in the following reaction: H+ + HCO3- → H2CO3 (CA)
• In acute respiratory alkalosis, for every 10 mmHg decrease in the PCO2, there is a 2meq/L decrease in the plasma HCO3- concentration.
• In chronic state renal compensation result in a 4 meq/L reduction in plasma [HCO3-] for every 10 mmHg reduction in PCO2. 25

26

Treatment of Respiratory Alkalosis
• Treat underlying cause• Breathe into a paper bag ???
• IV Chloride containing solution (hydrochloric acid, arginine chloride & ammonium chloride), Cl- ions replace lost bicarbonate ions
27

Metabolic Acidosis• Bicarbonate deficit - blood concentrations of
bicarb drop below 22mEq/L• Occurs when pH falls below 7.35• Causes:
– Loss of bicarbonate through diarrhea or renal dysfunction
– Accumulation of acids (lactic acid or ketones)– Failure of kidneys to excrete H+
28CO2 + H20 ↔ H2CO3 ↔ H+ + HCO3-
[H+] = 24 ×(PCO2 / [HCO3-])

Anion Gap Calculation• [Na+ + K+] – [Cl + HCO3] (Normal = 12 + 2) E.g :- Na 140, k 4 , CL 114, HCO3 18 (140 + 4) – (114 + 18) 144 – 132 = 12 normal E.g:- Na 140 , Cl 104, K 4.0, HCO3 10
(144) – (114) = 30 = High anion gap
Two types of Metabolic Acidosis– High Anion Gap = net gain of acid– Normal anion gap = loss of bicarbonate
29

High Anion Gap Differential(MUDPILES)
• Methanol• Uremia• DKA• Paraldehyde• Inborn Errors• Lactic
Acidosis• Ethylene
Glycol• Salicylates
Normal Anion Gap(USED CARP)
• Ureterostomy• Small bowel fistula• Extra Chloride• Diarrhea• Carbonic anhydrase
inhibitors• Addison’s disease• Renal tubular acidosis• Pancreatic fistulas Treatment: Replace
Bicarbonate30

Symptoms of Metabolic Acidosis• Headache, lethargy• Nausea, vomiting, diarrhea• Coma• Death
Compensation for Metabolic Acidosis• Increased ventilation• Renal excretion of hydrogen ions if possible• K+ exchanges with excess H+ in ECF( H+ into cells, K+ out of cells)
31CO2 + H20 ↔ H2CO3 ↔ H+ + HCO3
-

Compensation• Respiratory compensation results in a 1.2 mmHg
reduction in PCO2 for every 1.0 meq/L reduction in the plasma HCO3- concentration down to a minimum PCO2 of 10 to 15mmHg.
32
For example, if an acid load lowers the plasma HCO3- concentration to 9 meq/L, then:Degree of HCO3- reduction is 24 (optimal value) – 9 = 15.Therefore, PCO2 reduction should be 15 × 1.2 = 18.Then PCO2 measured should be 40 (optimal value) – 18 = 22mmHg.

33

Treatment of Metabolic Acidosis
• Treat the causes• Improve renal perfusion & acid excretion• NaHCO3, Dose = (weight Kg x base deficit x 0.3)• Ensure adequate ventilation
34

Metabolic Alkalosis• Characterized by
– Primary in HCO3 concentration greater than 26 mEq/L– Compensatory in PaCO2
• Classified according to urinary chloride– Chloride responsive– Chloride resistant
35

Metabolic AlkalosisChloride ResponsiveUrine Cl- < 20 mEq/L
Causes• Volume Contraction:
– Nasogastric suctioning, Gastric fistula– Vomiting , pyloric stenosis
• Post Hypercapnia• Low chloride intake• Hypomagnesemia• Penicillin
• Cystic fibrosis (sweat)• Alkali therapy (NaHCO3, Antacid abuse)• Chloride depletion (Diarrhoea & Diuretics
36

Metabolic AlkalosisChloride Unresponsive (resistant)
• Mineralcorticoid excess e.g Hyperaldosteronism• Exogenous steroids, Cushing’s disease• Alkali Ingestion• Licorice ingestion• Too much wine• Tobacco chewers• Bartter’s Syndrome
Urine Urine Cl- > 20 mEq/L> 20 mEq/L
37

Symptoms of Metabolic Alkalosis• Respiration slow and shallow• Hyperactive reflexes ; tetany• Often related to depletion of electrolytes• Dysrhythmias
Compensation for Metabolic Alkalosis• Alkalosis most commonly occurs with renal dysfunction, so can’t
count on kidneys• Alkali load• Acid loss - vomiting• Respiratory compensation difficult (hypoventilation limited by
hypoxia)
38

Compensation contn.• The development of alkalemia is sensed by central and
peripheral chemoreceptors, resulting in a reduction in the rate of ventilation and a reduction in tidal volume and thus an elevation in the pCO2.
39
pCO2 rises 0.7 mmHg for every 1.0 meq/L increment in the plasma [HCO3-].
For example, if an alkali load raises the the plasma HCO3- concentration to 34 meq/L, then:Degree of HCO3- elevation is 34 – 24 (optimal value)= 10.Therefore, PCO2 elevation should be 0.7 × 10 = 7
Then PCO2 measured should be 40 (optimal value) +7 = 47mmHg.

40

Treatment of Metabolic Alkalosis
• Electrolytes to replace those lost• Treat underlying disorder• IV chloride containing solution e.g saline (Chloride
Responsive)• Aldosterone antagonist (Chloride resistant)
41

Miscellaneous 1Arterial pH is related to the ratio of PCO2 to HCO3, both
pulmonary & renal compensatory mechanism are always such that PCO2 and HCO3 change in the same direction.
the exception occurs when there is a mixed acid base disorder. In that situation, multiple acid base processes coexisting may lead to a normal pH or a mixed picture especially when PCO2 & HCO3 moves in opposite direction
If the compensatory response is more or less than expected, by definition a mixed acid-base disorder exist.
42

An Alternative ApproachAn Alternative Approach that is rapid but perhaps less
precise is to correlate changes in Ph with changes in CO2 or HCO3.
• For a respiratory disturbance, every 10mmHg changes in CO2 should change arterial PH by approximately 0.08 U in the opposite direction.
• During metabolic disturbance, every 6mEq change in HCO3 also changes arterial PH by 0,1 in the same direction.
• If the change in pH exceed or is less than predicted, a mixed acid-base disorder is likely to be present.
• If the Arterial pH is relatively normal and the PCO2 and/or HCO3 are abnormal, one can assume that a mixed abnormality is present.
43

Miscellaneous 2• The Delta Ratio (∆/∆)• Assessment of elevated anion gap metabolic
acidosis to determine if a mixed acid base disorder is present.
44
Delta ratio = ∆ Anion gap/∆ [HCO3-] or ↑anion gap/ ↓ [HCO3-] = Measured anion gap – Normal anion gap Normal [HCO3-] – Measured [HCO3-]
= (AG – 12) (24 - [HCO3-])

Miscellaneous cont Delta ratio
Assessment Guidelines
< 0.4
Hyperchloremic normal anion gap acidosis
< 1
High AG & normal AG acidosis
1 to 2
Pure Anion Gap Acidosis Lactic acidosis: average value 1.6DKA more likely to have a ratio closer to 1 due to urine ketone loss
> 2
High AG acidosis and a concurrent metabolic alkalosisor a pre-existing compensated respiratory acidosis 45

CompensationPrimary DisorderPrimary Disorder Compensatory MechanismCompensatory Mechanism
Metabolic acidosisMetabolic acidosis Increased ventilationIncreased ventilation
Metabolic alkalosisMetabolic alkalosis Decreased ventilationDecreased ventilation
Respiratory acidosisRespiratory acidosis Increased renal reabsorption of HCOIncreased renal reabsorption of HCO33--
in the proximal tubulein the proximal tubule
Increased renal excretion of H in the Increased renal excretion of H in the
distal tubule distal tubule
Respiratory alkalosisRespiratory alkalosis Decreased renal reabsorption of HCODecreased renal reabsorption of HCO33--
in the proximal tubulein the proximal tubule
Decreased renal excretion of HDecreased renal excretion of H++ in the in the
distal tubule distal tubule 46

Primary disorder
Initial chemical change
Compensatory
response
Compensatory
Mechanism
Expected level of compensation
Metabolic Acidosis
↓HCO3- ↓PCO2 Hyperventilation
PCO2 = (1.5 × [HCO3-]) + 8 ± 2 ↓PCO2 = 1.2 ×∆ [HCO3-]
Metabolic Alkalosis
↑HCO3- ↑PCO2 Hypoventilation PCO2 = (0.9 × [HCO3-]) + 16 ± 2 ↑PCO2 = 0.7 × ∆ [HCO3-]
Respiratory Acidosis
↑PCO2 ↑HCO3-
Acute Intracellular Buffering (hemoglobin, intracellular proteins)
↑[HCO3-] = 1 mEq/L for every 10 mm Hg ∆PCO2
Chronic
Generation of new HCO3- due to the increased excretion of ammonium.
↑[HCO3-] = 3.5 mEq/L for every 10 mm Hg ∆PCO2
Respiratory Alkalosis
↓PCO2 ↓HCO3-
Acute Intracellular Buffering ↓[HCO3-] = 2 mEq/L for every 10 mm Hg ∆PCO2
Chronic
Decreased reabsorption of HCO3-, decreased excretion of ammonium
↓[HCO3-] =4 mEq/L for every 10 mm Hg ∆PCO2
47

Acidemia (PH<7.35)
PCO2
High NormalOr low
Normalor high
Incompatible [HCO3][HCO3]
High Normal Low Low
Chronic respiratory acidosis
Acute respiratory acidosis
Mixed respiratory and metabolic acidosis
Metabolic acidosis48

↓ Plasma [HCO3]
Anion gap
Normal Increased
High anion gap metabolic acidosisPlasma [K]
Low Normal High
Hypokalemic hyperchloremic metabolic acidosis
Respiratory alkalosis
Hyperkalemic hyperchloremic metabolic acidosis49

Alkalemia (PH>7.45)
PCO2
Low High\Normal
Normalor
Low Incompatible
High Normal
[HCO3]
Low High
[HCO3]
Mixed respiratory and metabolic alkalosis
Acute respiratory alkalosis
Chronic respiratory alkalosis
Metabolic alkalosis
50

↑ Plasma [HCO3]
Metabolic alkalosis Respiratory acidosis
Spot urine [Cl]
>20mmol\L <20mmol\L
Saline unresponsive metabolic alkalosis
Saline responsive metabolic alkalosis
51

Example• A patient is in intensive care because he suffered
a severe myocardial infarction 3 days ago. The lab reports the following values from an arterial blood sample:– pH 7.3– HCO3- = 20 mEq / L ( 22 - 26)– pCO2 = 32 mm Hg (35 - 45)
Diagnosis• Metabolic acidosis• With partial compensation
52

CASE 1
• A 44 year old moderately dehydrated man was admitted with a two day history of acute severe diarrhea. Electrolyte results: Na+ 134, K+ 2.9, Cl- 108, HCO3- 16,
• Urea 31, Cr 1.5.•
ABG: pH 7.31 pCO2 33 mmHg HCO3 16 pO2 93 mmHg
53

CASE 2
• A 22 year old female with type I DM, presents to the emergency department with a 1 day history of nausea, vomiting, polyuria, polydypsia and vague abdominal pain. P.E. noted for deep sighing breathing, orthostatic hypotension, and dry mucous membranes.
• Labs: Na 132 , K 6.0, Cl 93, HCO3- 11 glucose 720, Urea 38, Cr 2.6. UA: pH 5, SG 1.010, ketones negative, glucose positive . Plasma ketones trace. ABG: pH 7.27 HCO3- 10 PCO2 23
• What is the acid base disorder?
54

CASE 3
• A 70 year old man with history of CHF presents with increased shortness of breath and leg swelling.ABG: pH 7.24, PCO2 60 mmHg, PO2 52 HCO3- 27
• What is the acid base disorder?
55
![A genetic origin for acid–base imbalance triggers the ......A genetic origin for acid–base imbalance triggers the mitochondrial damage that explains the… 53 13 receptor2(SSTR2)[5].Tumorprogressionisclassically](https://static.fdocuments.in/doc/165x107/601b91f8e5079e601b352dc4/a-genetic-origin-for-acidabase-imbalance-triggers-the-a-genetic-origin.jpg)