A large randomised controlled trial among trauma patients with significant haemorrhage, of the...
-
Upload
vernon-hensley -
Category
Documents
-
view
217 -
download
1
Transcript of A large randomised controlled trial among trauma patients with significant haemorrhage, of the...
A large randomised controlled trial among trauma patients with
significant haemorrhage, of the effects of antifibrinolytic treatment on
death and transfusion requirement
Global deaths at ages 5-45 years (both sexes)
Global deaths at ages 5-45 years (both sexes)
Deaths (2000 projected)
HIV/AIDS 2,104,454
Road traffic injury 657,614
Tuberculosis 603,522
Self inflicted injury 431,924
Violence 335,202
War injuries 167,329
Exsanguination CNS injury
Organ failureOther
45% 41%
10%4%
In-hospital trauma deathsIn-hospital trauma deaths
Sauaia A et al. Epidemiology of trauma deaths: a reassessment. J Trauma 1995;38:185-193
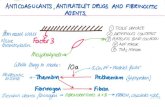
FibrinolysisFibrinolysis
• During fibrinolysis, plasminogen is converted into plasmin by tissue plasminogen activator
• Plasmin binds to fibrin via its lysine binding sites to cause fibrinolysis.
Pharmacologic inhibition of fibrinolysis
• Tranexamic Acid (TXA) is a synthetic derivative of the amino acid lysine.
• It has a very high affinity for the lysine binding sites of plasminogen.
• It blocks these sites and prevents binding of activated plasminogen to the fibrin surface, thus exerting its antifibrinolytic effect.
SYSTEMATIC REVIEWS
APROTININ61 Randomised controlled trials including 7,027 participants
TRANEXAMIC ACID (TXA)18 Randomised controlled trials including 1342 participants
Antifibrinolytic agents in elective surgery
Henry DA et al. Antifibrinolytic use for minimising perioperative allogeneic blood transfusion (Cochrane Review). In: The Cochrane Library, Issue 3, 2004. Chichester, UK: John Wiley & Sons, Ltd
NEED FOR TRANSFUSION
TXA
Aprotinin
Antifibrinolytic better Antifibrinolytic worse
0.70 (0.64-0.76)
0.66 (0.54-0.81)
RR (95% CI)
0 0.4 0.8 1.2 1.6 2.0
Antifibrinolytic agents in elective surgery
Henry et al
BLOOD UNITS SAVEDBLOOD UNITS SAVED
0 0.4 0.8 1.2 1.6 2.0
Blood units
1.1 (0.7-1.5)
1.0 (0.7-1.4)TXA
Aprotinin
Antifibrinolytic agents in elective surgery
Henry et al
RE-OPERATIONRE-OPERATION
RR (95% CI)
Antifibrinolytic better Antifibrinolytic worse0 0.4 0.8 1.2 1.6 2.0
TXA
Aprotinin 0.40 (0.25-0.66)
0.72 (0.29-1.79)
Antifibrinolytic agents in elective surgery
Henry et al
MORTALITYMORTALITY
TXA
Aprotinin
RR (95% CI)
Antifibrinolytic better Antifibrinolytic worse0 0.4 0.8 1.2 1.6 2.0
0.87 (0.63-1.19)
0.43 (0.15-1.18)
Antifibrinolytic agents in elective surgery
Henry et al
Tranexamic acid for minimising surgical blood loss
Henry et al
ADVERSE EFFECTS
RR 95% CINon-fatal MI 0.69 0.21 - 2.29Stroke 2.27 0.65 - 7.99DVT 0.84 0.30 - 2.30PE 0.32 0.07 - 1.56Any thrombosis 0.98 0.49 - 1.94
No evidence of increased adverse effects
TXA already being used in some treatment protocols
• Crystalloids (warm) 1L (250X4)• Blood -2 units + Tranexamic acid IV 2gr• rFVIIa 9.6 mg• Blood -2 units, consider BTB transfusion • Fibrinogen + bicarbonate+ rFVIIa 9.6mg • Cont. fluids + blood according patient’s condition • Repeat rFVIIa in case of re-bleeding
Treatment protocol of Israeli Defence Force
1 randomised controlled trial including 70 patients
Drug versus Placebo: 0 vs. 3 deaths
Roberts IG, Coates T, Shakur H Antifibrinolytic drugs for acute traumatic injury. Cochrane Database Syst Rev. 2004 Oct 18(4):CD004896
Antifibrinolytic agents in trauma
Systematic review
• Insufficient evidence to support clinical use in trauma
• Need for RCT – CRASH-2
• Insufficient evidence to support clinical use in trauma
• Need for RCT – CRASH-2
• Bleeding is a leading cause of trauma death • Blood transfusion can be dangerous• Antifibrinolytics reduce blood loss after
surgery• Surgery and trauma produce similar
haemostatic responses• Trials in trauma too small to confirm or
refute a moderate effects• A simple intervention like TXA could prevent
thousands of trauma deaths and transfusion associated infections
• MRC CRASH trial showed that large numbers of patients can be enrolled in the emergency setting
Rationale for CRASH-2
Rationale for TXA
• TXA may be as effective as aprotinin*
• Lower cost of TXA
• Need for a test dose of aprotinin to assess for potential allergic reactions (not practical in emergencies)
• In some settings TXA more acceptable than aprotinin which is derived from bovine lung
* Henry et al
Aims
• To quantify the effect of tranexamic acid (TXA) on death and transfusion requirement in adult trauma patients with significant ongoing haemorrhage, or who are considered to be at risk of significant haemorrhage
• To quantify the effect of such treatment on the risk of non-fatal vascular events
DOCTOR IS “REASONABLY CERTAIN” THAT ANTI-FIBRINOLYTIC AGENTS
ARE INDICATED.
INELIGIBLEGIVE ANTI-FIBRINOLYTIC AGENTS;
DO NOT RANDOMISE.
DOCTOR IS “REASONABLY CERTAIN” THAT ANTI-FIBRINOLYTIC AGENTS ARE CONTRA-INDICATED.
INELIGIBLEDON’T GIVE ANTI-FIBRINOLYTIC AGENTS;DO NOT RANDOMISE.
Doctor is “SUBSTANTIALLY UNCERTAIN” as to the appropriateness of
anti-fibrinolytic agents in this patient
TELEPHONE FOR RANDOMISATION OR PAPER RANDOMISE
TRANEXAMIC ACID PLACEBO
POTENTIALLY ELIGIBLE
Trauma patients judged to be 16 years or older, with significant haemorrhage (systolic blood pressure less than 90 mmHg and/or heart rate more than 110
beats per minute), or considered to be at risk of significant haemorrhage, within 8 hours of the injury
What is meant by ‘risk of significant haemorrhage’
• Patients with major trauma who are likely to need an early blood transfusion in the view of the attending doctor after taking into account mechanism of injury, findings from secondary survey, physiology and response to fluid infusion
Randomisation
• Give brief patient details
• Computer determines treatment allocation
• Treatments ‘balanced’ on prognostic factors
• Select lowest numbered treatment pack
• Patient details sent by via secure website, email or fax
Central (by telephone)Central (by telephone)
Non-centralNon-central
• CRASH-2: involve patients who have suffered serious injuries and are at risk of life threatening haemorrhage
• Most patients will have some impairment in their level of consciousness caused either by blood loss or coexisting head-injury
• Patients may not be able to provide written informed consent
• Trial treatment has to be administered as soon as possible after injury
• Need to comply with local approved consent process
Consent
Patient Entry
PATI ENT ENTRY ALL QUESTIONS BELOW NEED TO BE ANSWERED BEFORE CALLING THE RANDOMISATION SERVICE
INFORMATION ABOUT YOUR HOSPITAL
1. Country
2. Name of hospital (or your hospital code)
3. Name of caller
INFORMATION ABOUT THE PATIENT
4. Patient sex (please circle) Male Female 5. Patient initials
6. Patient hospital identification number
7. Do you know patient’s date of birth?
a. YES – date of birth / / b. NO – approximate age
INFORMATION ABOUT THE INJURY
8. Estimated number of hours since injury hours
9. Type of injury (please circle) 1 Blunt 2 Penetrating 3 Both
FIRST MEASUREMENT IN HOSPITAL OF THE FOLLOWING ( IF UNKNOWN GIVE VALUE AT RANDOMISATION)
10. Systolic BP (mmHg) 11. Respiratory rate (per min)
12. Central capillary refill time (sec) 13. Heart rate (per min)
EYE OPENING MOTOR RESPONSE VERBAL RESPONSE 4 Spontaneous 6 Obeys commands 5 Orientated
3 To sound 5 Localising 4 Confused speech
2 To pain 4 Normal flexion 3 Words
1 None 3 Abnormal flexion 2 Sounds
2 Extending 1 None
14. Glasgow Coma Score (max 15)
1 None
Now call Randomisation Service with these answers and write down the treatment pack number given at the end of the phone call
Box Pack Get this pack and follow the instructions on it carefully
Or paper randomise as per instructions in site file
Please complete before randomisation to ensure patient fulfils eligibility criteria
Please complete before randomisation to ensure patient fulfils eligibility criteria
TreatmentAmpoule
s
Dose (Tranexamic Acid
or placebo)Infusion rate
Loading 2 1 gram100 ml over 10 minutes
Maintenance
2 1 gram
120 mg/hr [60 ml/hr] for about 8 hours
Treatment
Fixed dose more practicable in emergency situation
Dose within range shown to inhibit fibrinolysis and provide haemostatic benefit
Fixed dose more practicable in emergency situation
Dose within range shown to inhibit fibrinolysis and provide haemostatic benefit
Treatment
EACH BOX WILL CONTAIN 8 PATIENT TREATMENT PACKS
Each pack contains:4 ampoules of tranexamic acid/placebo100 mL bag sodium chloride1x 10 mL syringe1x green needle
Plus:Adhesive labels for medical notes and data formsConsent forms (if needed)Patient information sheetData forms (Entry & Outcome)
Outcome data
OUTCOME FORM COMPLETE AT DISCHARGE FROM THE RANDOMISING HOSPITAL,
DEATH IN HOSPITAL OR 28 DAYS AFTER INJURY, WHICHEVER OCCURS FIRST
1. HOSPITAL
2. PATIENT Patient Initials Hospital I D Number Sex M F
Date of Birth DAY / MONTH / YEAR .
3. OUTCOME 3.2 PATIENT ALIVE
…. Discharged – Date of discharge DAY / MONTH/ YEAR.
…. Still in this hospital now (28 days after inj ury) – Date DAY / MONTH/ YEAR..
3.3 IF ALIVE TICK ONE BOX THAT BEST DESCRIBES THE PATIENT’S CONDITION ( at 28 days or prior discharge)
3.1 DEATH IN HOSPITAL
Date of death DAY / MONTH/ YEAR.
Cause of death
…. Bleeding
…. Head injury
…. Myocardial Infarction
…. Stroke
…. Pulmonary Embolism
…. Multi organ failure
…. Other – describe
…………………………………
…. No symptoms
…. Minor symptoms
…. Some restriction in lifestyle but
independent
…. Dependent, but not requiring constant
attention
…. Fully dependent, requiring attention
day and night
4. MANAGEMENT 7. TRANSFUSION
a) Days in I ntensive Care Unit (if not admitted to I CU, write ‘0 ’ here)
a) Blood products transfusion YES NO
b) Significant Head I njury YES NO b) Units transfused in 28 days
c) Operation site - Tick one box on every line Red cell products units
Neurosurgical YES NO Fresh frozen plasma units
Chest YES NO Platelets units
Abdomen YES NO Cryoprecipitate units
Pelvis YES NO
Recombinant Factor VI Ia YES NO
5. COMPLICATIONS 8. PERSON COMPLETING FORM
Tick one box on every line NAME
Pulmonary Embolism YES NO POSITION
Deep Vein Thrombosis YES NO DATE
Stroke YES NO
Operation for bleeding YES NO
NOW SEND THIS FORM TO THE CO-ORDINATING
CENTRE IN ONE OF THE FOLLOWING WAYS:
Myocardial Infarction YES NO SECURE WEBSITE
Gastrointestinal bleeding YES NO
ELECTRONIC DATA FORMS / EMAI L
6. TRIAL TREATMENT FAX +44 (0)20 7299 4663
a) Complete loading dose given YES NO
b) Complete maintenance dose given YES NO
(Hospital name or code)
Attach treatment
pack sticker here
Primary outcome measure: death in hospital within four weeks of injury
Cause of death is described to assess whether due to haemorrhage or vascular occlusion.
Primary outcome measure: death in hospital within four weeks of injury
Cause of death is described to assess whether due to haemorrhage or vascular occlusion.
CRASH Trials Co-ordinating CentreLondon School of Hygiene & Tropical Medicine
Keppel Street, London WC1E 7HT
Tel +44(0)20 7299 4684, Fax +44(0)20 7299 4663Email [email protected]
WWW.CRASH2.LSHTM.AC.UK WWW.CRASH2.LSHTM.AC.UK