20320140506005 2
-
Upload
iaeme -
Category
Technology
-
view
37 -
download
0
Transcript of 20320140506005 2

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
37
EVALUATION OF THE DRINKING WATER QUALITY AND THE
EFFICIENCY OF AL-HAWIJA WATER TREATMENT PLANT: A CASE
STUDY IN IRAQ
Rodhan Abdullah salih
Al-Hawija Technical Institute, Foundation of Technical Education/Iraq
ABSTRACT In this paper laboratory tests measured the concentrations of some of the physical, chemical and biological properties to assess the efficiency of Al- Hawija water treatment plant and the quality of drinking water. Samples were taking of raw and treated water .The process repeated every month and for the period of November 2013 -April 2014. The results of physical and chemical examination of raw water are within the limits of the standard specifications of the World Health Organization (WHO). Experimental based evaluation work with efficient final results. Research work ensures the quality of drinking water. The evaluation of the efficiency of the stages of conventional treatment showed that the plant operated by efficiently (65.97) % of sedimentation stage. Filtration efficiently of the plant ranged between (59-75 %).The efficiency of sterilization is good except for the month of November 2013 (86%) and the months of March and April 2014 (80% and 82 %), respectively. SPSS statistical program used for the purpose of conducting statistical analyzes of the characteristics of the raw and treated water, and found a positive correlation(R) between some of significantly characteristics. Key word: Efficiency, Treatment Plant, Quality, Water.
1-INTRODUCTION Iraq faced dozen of years since a significant lack of services provided to citizens and comes on top of these services, the processing of drinking water quality and conforms to the specifications of World Records. To achieve this purpose, drinking water treatment plants, should be characterized by high efficiency. Allaa M. Aenab, S.K. Singh [1] indicates that 80 % of Iraq does not treat their water before drinking. Drinking water is a natural water where there are standards and determinants
INTERNATIONAL JOURNAL OF CIVIL ENGINEERING
AND TECHNOLOGY (IJCIET)
ISSN 0976 – 6308 (Print)
ISSN 0976 – 6316(Online)
Volume 5, Issue 6, June (2014), pp. 37-54
© IAEME: www.iaeme.com/ijciet.asp
Journal Impact Factor (2014): 7.9290 (Calculated by GISI)
www.jifactor.com
IJCIET
©IAEME

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
38
of physical, chemical and bacteriological to the World Health Organization, whether a piece of natural source like a river or spring, or after a purification [2] .Drinking water was a water that can be addressed through drinking by humans [3, 4], or a water of sufficient quality to serve as drinking water is potable water or can be defined as the water is not harmful to humans or non-polluting pretty unhealthy [5]. Water resources suffer of many changes of physical, chemical and biological characterize, and these changes lead to contamination of the water. Iraq government institutions in to conduct measurements to examine the levels of dissolved solids and metal elements tests as one of the basic criteria for measuring the quality of surface water and the variables that are going to the Tigris and Euphrates, One of these tests: - Cl, sulfates So4, total dissolved solids TDS and other tests and these tests are considered part of the indicators of water quality and the level of suitability for human use.
2-PREVIOUS STUDIES
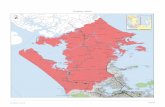
The researcher Khalid Mohammed Shaheen[6]conduct an assessment study of AL-Qubba water treatment plants (AL- Mosul city, Iraq ).He was compared the results of chemical and physical tests of raw and treated water with the standard specifications of the World Health Organization. The study showed good efficiency of the plant in removal of turbidity and suspended solids, in spite of the lack of efficiency acceptable to remove hardness and dissolved solids .The researcher showed why the piece does not contain the station concerned and means to facilitate the water, and also to the excessive use of aluminum sulfate (alum.) in the process of coagulation. Lower deposition efficiency because of PH values was greater than the values appropriate to the interaction o aluminum sulfate and this leading to ionization of aluminum hydroxide and dissolution in water. Performance of Al-Dewanyia Water Treatment Plant in Iraq [7] is an essential parameter to be monitored and evaluated to better understanding of design and operating difficulties in water treatment plants. This evaluation may determine required recommendations and highlight modification requirements for continuous design and operating schemes. The evaluation was carried out by reviewing the engineering design to assure matching of standards and codes. Also, physical turbidity and TSS (total suspended solids), and biological, analysis were conducted to investigate water quality. Treated water is not conforming to (WHO). Ali Hammed Mohammed; Alaa A.Shakir [8] evaluated the performance of AL-wahdaa project drinking water treatment plant, Baghdad, Iraq. In this study, the removal efficient of filtration; sedimentation will be addressed as well as the turbidity during three years of investigation. It was found that the average value of the removal of sedimentation was about 46% (R ² = 0.902) and the rate of filtration efficiency 75% (R ² = 0.445).The total rate of turbidity values was (18 NTU) which is more than the permissible limits according to WHO standards, and high values TDS (total dissolved solids), especially during the month of January due to high turbidity discharge. This research aims to study the efficiency of the stages of conventional treatment (sedimentation, filtration and disinfection) and the water quality of Hawija water treatment plant in the city of Hawija (One of the largest cities of Kirkuk governorate, Iraq ) 70 km to the southwest of the governorate. The number of beneficiaries from the station about 88 thousand people [9]. It is one of the biggest stations in the city. It was constructed in 2004 .The total area of the station is (50000) m ² and a design capacity of 1833 m³ / h [9]. The station supplied with water from the AL-Hawija River and coordinates the global system (E = 43˚46'10 "), (N = 35˚ 20' 15") as shown in Fig. (1).

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
39
Fig.1: An Aerial Photograph of AL-Hawija Water Treatment Plant
3-DESCRIPTION OF THE PLANT
3.1. Intake It is located on AL-Hawija water table in the depth of 5 meter, a rubber protector to prevent the entry of floating material. The Low Lift Pump contains 4 plugs on the pump uploaded the raw water, 3 of them are working and the one –pump is standby. The discharge of each pump is 700m³/h; the head pressure of water is 4m. These pumps are meant to raise the water from the river to the Rapid mixing basin. .
3.2. The Purification Process
3.2.1 The rapid mixing Process is the first treatment step for water after receives it from intake. Aluminum Sulfate added and is mix ing with raw water by flash mixer. 3.2.2 Sedimentations Basins Water entry from basins mixing speed directly to the clarifier the first of which is (20 m*22 m)and has sweeping clays at the bottom and the installation from the top makes it work sweeping clays floating and make up on this machine in practice moves water then to the basin sedimentation, who has the same dimensions of the basin the first but does not contain sweeping clays and both basins of the depth of each and middle are grouped clays in the medium is pulled by located in the center for this purpose. 3.2.3 Filtration Basins Water is pumped from the basin of sedimentation to the second filter presses that are working on the plant, as there are ten filters; the area of each filter is 40 m². 3.2.4 Disinfection stage After the withdrawal of water from the filters passes to the serialization room which located behind filters where added chlorine and other disinfectants before the passage of water into a reservoir of treated water.

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
40
3.2.5 The flocculation stage is the second step in purifying the raw water which is held in the basin of flocculation which is round basin contains inanimate to increase the surface area. It is of concrete column which is medley .Raw water is mixed with aluminum sulfate at the bottom of the basin, then the water goes into the sintering after a period of 30 minutes and then to sedimentation basins. The duration time of water or that the water remains in sedimentation basins is (2-4) hours. Fig. (2): the basic components of AL- Hawija water treatment plant.
Fig.2: the basic components of AL- Hawija water treatment plant.
4. DATA PROCUREMENT
4.1-Sampling collection The samples were collected for the examination of physical and chemical properties by the plastic cans (one-liter).Samples were taken from the raw and treated water. The physical and chemical properties of the samples were conducted by the equipment available in the laboratories of the Department of Water Resources /AL- Hawija Technical Institute, Kirkuk /Iraq, as one sample every month. Turbidity was measured using a device (micro 100 IR Turbidity Meter), while the PH, EC, TDS, sulfates and nitrates have been measured by using a device (Multi-Parameter PCS Tester Tm 3s). Alkanets, Chloride, Total Hardness, calcium, Magnesium were measured by titration. For Biological examination, samples were collected by sterile plastic bottles, for the period from November 2013 to April 2014, and tested according to standard methods adopted in the collection, preservation and analysis of samples [10]. Column deposition is made for the purpose of estimating the efficiency of sedimentation, which is a tube of aluminum in diameter (200 mm) and height (2.5 m); with openings for sampling at different heights from the base which is (0.5 0.1, 1.5 0.2, 2.5 m) as shown in Fig.3
1-Well clouds
2-The low lift pumps3-Well assembly
4-Sedimentation basins5-Filters
6-Ground Reservoir7-Pumps Lifting Higher8-Sediment tank assembly
9-Higher reservoir
To the
dis
trib
ution
netw
ork
1-Well suction
2-The low lift pumps3-Well assembly
4-Sedimentation basins5-Filters
6-Ground Reservoir7- High Lift Pumps8-Sediment tank assembly
9-Higher reservoir
10 11
1-Well suction
2-The low lift pumps
3-Well assembly
4-Sedimentation basins
5-Filters6-Ground Reservoir
7- High Lift Pumps
8-Sediment tank assembly
9-Higher reservoir
10-Aluminum sulfate House
11-Chlorination house

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
41
Fig.3: Sedimentation column
Samples were taken from the raw water and was measured the concentration of initial suspended solids (Co) in units of mg / l. Sedimentation column was full with raw water from rapid mixing basin which has a concentration of homogeneous suspended solids through various depths. Samples was taken from the same depths and at different time periods that represented the detention time (1, 2, 4) an hour and then calculated a concentration of particles that are less than the speed of sedimentation h/t, where h = the depth at which it took the form, t = period of sedimentation [11, 12].These results can be plotted curves of changes in the distribution of sedimentation as given in Figure 4.
Fig.4: Curve of analysis sedimentation for free particle.
ةعرس بيسرتلا v
لا ذمكرو
م نقا لس رع ةوذ
لاجسيامت
ن سب=ة
cF
ractio
n w
ith
se
ttin
g v
elo
city less th
an
sta
ted
Velocity mm/sec

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
42
The rate of overflow Q = Vs A, where Vs= velocity of sedimentation, A=area, the molecules that have a speed greater than the Vs sedimentation will remain entirely. The ratio of the particle to be removed (1-CO) as: - CO = the proportion of the remaining particles, the ratio of the particle sedimentation velocity, which has less than Vs be V/Vs [11, 12]. Therefore, the concentration of particles to be removed can be found from the following equation [12]: -
-------------------- (1)
Total removal of water is: -
∫+−=oC
s
o vdcV
CC0
1)1( ------------------------- (2)
Extent of integration in the equation (2) above can be found from the graph or use approximate theory or the theory of Newton Simpson _ Rap soon.
4.2- Estimation the efficiency of sand filters When the water is passing through a layer of sand containing suspended solids, much of the loose material will be removed, although removal of these materials get into the pores due to a combination of physical and chemical processes, and is estimated efficiency of the filters by finding total suspended solids before water is entering the filters and after it is leaving them and by applying the following equation [13, 14]: - Efficiency filters% = 1 - (total suspended solids of the water out of the filters / total suspended solids of water prior to their entry to the filters) * 100. --------------- --- (3)
4.3-Estimation the efficiency of sterilization Water Sterilization is to kill microorganisms that cause disease that the water contains, and chlorine used widely in water sterilizing. Bacteria and pathological material needed to be sterilized but non-toxic to humans. Specifications of the World Health Organization (WHO) considers that the water is drinkable if it contained the number of total bacterial cells less than 50 in 100 ml and was free of E.coli. The efficiency of sterilization can calculate of the following equation [15]: Sterilization efficiency% = 1 - (number of pathogenic bacteria for treated water / number of Pathogenic bacteria of raw water) 100---------------------- (4)
5-CALCULATIONS AND RESULTS
5.1-Sedimentation efficiency After collecting samples from the sedimentation column and at different depths and at different times, suspended solids were calculated which remains in each sample and the results as shown in table (1)
∫oc
s
VdcV
0
1

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
43
Table (1): the concentration of suspended solids in the sedimentation basin
Estimation settling velocity and the percentage of materials that have suspended speed Sedimentation less than the designated speed and the results were as shown in table (2).
Table (2): speeds calculations and the suspended solids%
remaining material %
removed
%material Speed
)mm / sec( Time (sec) Depth (mm)
58 42 0.139 3600 500
39 61 0.069 7200 500
42 58 0.035 14400 500
55 45 0.277 3600 1000
37 63 0.138 7200 1000
35 65 0.069 14400 1000
79 21 0.416 3600 1500
65 35 0.208 7200 1500
51 49 0.014 14400 1500
79 21 0.556 3600 2000
65 35 0.278 7200 2000
58 42 0.138 14400 2000
86 14 0.674 3600 2500
77 23 0.347 7200 2500
65 35 0.174 14400 2500
Draw a curved distribution of total suspended solids in the water by the percentage of total suspended solids in the liquid out of the sedimentation basin with sedimentation velocity, as shown in Fig. 5. Discharge of each sedimentation basins = Design plants discharge / number of basins – (5) = 1833/3 = 611 m³ / h = 0.169 m ³ / sec Sedimentation basin area = (15) ² × 3.14 = 706.5 m ² Design speed (Vs) = Discharge / Area ---------------------- (6) = 0.169 / 706.5 = 0.000239 m / sec = 0.239 mm / sec. From the distribution curve for the remaining suspended solids and design speed (Vs =0.239), we can calculate the remaining suspended solids corresponding to the design speed which is equal to 69%, which represents the (Co) in the equation (2). The efficiency of the sedimentation basin calculated by the equation of removal , after finding space between the curved distribution of total
Depth of the sample
(m)
The concentration of suspended solids (mg / L)
Detention time (time of sedimentation) / h
0 1 2 4
0.5 396 168 244 232
1 396 180 250 257
1.5 396 83 139 192
2 396 83 138 166
2.5 396 55 91 138

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
44
suspended materials and the y-axis; and the horizontal line that passes the value of (Co) of the curve which is equal to (8.36) as follows: - E %= (100 – 69) + (1/0.239) * 8.36 = 65.97 %
Fig.5: Distribution curve for residual suspended solids
5.2-Filters Efficiency Total suspended solids (TSS) for the water before and after filters is calculated and filters efficiency is computed by applying equation (3) as listed in Table 3.
%Table 3: shows the TSS of water outside and inside of the filters and filtration efficiency
%Filters efficiency TSS of water outside filters
TSS of water entering the filters
Month
64 20 56 November
75 16 66 December
59 18 44 January
63 33 90 February
71 23 80 March
69.7 26 86 April
61 28 72 May
67.3 17 52 June
It is clear from these results that the filtration efficiency ranged between 59% in the month of January and 75% in the month of December.
5.3-The efficiency of sterilization Table (4) and figs (6), (7) and (8) shows the biological properties of water plant and free residual chlorine and sterilization efficient of the samples taken from the plant for the duration of the study, and determine the total number of bacteria and pathogenic bacteria. The efficiency of sterilization can by calculated applying equation (4). Plant efficiency in the treatment of E-coli(

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
45
pathogenic) bacteria was good throughout the study period, except for the month of November 2013 (86%), this is due to the broken sewer pipes and water mixing of water network through some of the defects and shortcomings occurring; and the months of March and April 2014 (80%, 82%), respectively so as not to add chlorine gas. Sterilization process stopped once and for the period of early 2014 and until mid-April of the same year .The security authorities is lifting the bottles of chlorine gas from the station once and for unknown reasons.
Table (4): biological characteristics and free residual chlorine and sterilization efficiency Sterilization efficiency %
E-coli for treated water / 100 ml
E-coli for raw water /
100 ml
Total bacteria treated
water / 100 ml
Total bacteria raw water / 100
ml
Free chlorine mg / l
Month
86 10 70 30 50 1.1 Nov.
100 0 100 40 100 2.6 Dec. 100 0 50 50 200 2.8 Jan. 100 0 32 35 100 2.5 Feb. 80 20 100 90 250 --- March 82 5 28 60 180 --- April
Fig.6: The total number of bacteria
Fig.7: The number of E-coli (pathogenic) bacteria
0
50
100
150
200
250
300
Nov Dec Jan Feb March Apr
Months
To
tal
ba
cte
ria
raw water
Treated water
0
20
40
60
80
100
120
Nov Dec Jan Feb March Apr
Months
Path
agonic
bacte
ria
raw water
treated water

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
46
Fig.8: Plant efficiency for E-coli( pathogenic) bacteria
5.4-Water quality The physical and chemical characteristics for raw and treated water were examined including temperature, turbidity, PH, electrical conductivity, Alkanets, total hardness, calcium, magnesium, chloride, sulfate, TDS, suspended solids and nitrates and listed in tables (5) and (6).
Table (5): physical and chemical characteristics of the raw water Month Tem.
C˚ Tur. NTU
PH E.C µm/cm
ALK. mg/l
T.H mg/l
Ca mg/l
Mg mg/l
Cl mg/l
So4 mg/l
TDS mg/l
TSS mg/l
No3 mg/l
Nov 19 50 8.2 480 115 304 75 70 161 43 380 33 2.57 Dec 12 23 8.1 423 175 307 56 51 154 98 595 20 3.8 Jan 10 33 7.9 409 139 350 80 40 196 83 677 37 3.75 Feb 9 39 8.1 490 201 300 102 51 183 46 353 67 2.77
March 16 25 8 430 100 308 105 52 266 107 371 160 3.6 Apr 26 19 7.9 456 117 303 120 60 263 127 599 143 3.89 May 30 15 8 470 145 308 125 63 219 109 583 31 2.38
Average 17.4 29.1 8 451 141.7 311.4 94.7 55.2 206 87.5 508.2 70.1 3.2
Table (6): physical and chemical characteristics of the treated water
Month Tem. C˚
Tur. NTU
PH
C E. µm/cm
ALK.
mg/l
T.H mg/l
Ca mg/l
Mg mg/l
Cl mg/l
So4 mg/l
TDS mg/l
TSS mg/l
No3 Mg/
l Nov 20 3.6 7.9 476 111 301 72 64 143 40 372 31 2.4 Dec 13 4.9 7.8 418 160 362 51 48 123 93 567 18 3.7 Jan 11 6.5 7.7 407 126 335 78 33 174 78 671 35 3.65 Feb 10 5.6 8 482 196 295 93 47 166 39 348 63 2.4
March 18 3.9 7.9 426 143 298 97 49 240 98 366 57 3.2 Apr 25 2.3 7.8 448 108 296 111 54 245 117 574 41 3.63 May 31 5.5 7.6 466 136 290 117 57 209 89 560 28 2.15
Average 18.2 4.6 7.8 446.1 140 311 88.4 50.2 185 79 494 42.5 3.01 WHO - 5 6.5
-8.5
1600 250 500 150 100 250 250 1000 - 50
These tables show that all the properties of the treated water within the international standard specifications WHO [16], and that the advantage of the characteristics of the raw water quality is good. Figs (9-19) describe the variation of physical and chemical characteristics of the raw and treated water with time.
0
20
40
60
80
100
120
Nov Dec Jan Feb March Apr
Months
Effic
ien
cy%
Treated water

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp.
Fig.9: Variation of Turbidity with time
Fig.
Fig.
0
10
20
30
40
50
60
Nov
Turb
idity (N
TU
)
International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976
6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
47
9: Variation of Turbidity with time
Fig.10: Variation of PH with time
Fig.11: Variation of E.C with time
Dec Jan Feb March Apr May
Time (month)
Raw water
Treated water
International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
May

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp.
Fig.12: Variation of ALK with time
Fig.
Fig.
International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976
6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
48
12: Variation of ALK with time
Fig.13: Variation of T.H with time
Fig.14: Variation of Ca with time
International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp.
Fig.
Fig
Fig.
International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976
6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
49
Fig.15: Variation of Mg with time
Fig.16: Variation of Cl with time
Fig.17: Variation of So4 with time
International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp.
Fig.18: Variation of TDS with time
Fig.
6-STATISTICAL ANALYSIS OF THE RESULTS
SPSS statistical program was used for the purpose of conducting statistical analysis of the characteristics of the raw and treated water, and found a positive correlation significantly among some of the physical and chemical characteristics.correlation(R) of the raw and treated water. Table (7) illustrates the interdependence of the raw water. Table (8), which illustrates the interdependence of the treated water. A psignificant can be seen among some of the physical and chemical characteristics, but the high value of a positive correlation of raw water can be seen between Ca and Cl(R=0.8510. A positive correlation significant of treated water can 0.874).
International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976
6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
50
18: Variation of TDS with time
Fig.19: Variation of No3 with time
STATISTICAL ANALYSIS OF THE RESULTS
SPSS statistical program was used for the purpose of conducting statistical analysis of the characteristics of the raw and treated water, and found a positive correlation significantly among
physical and chemical characteristics. Tables (7) and (8) show the matrix of correlation(R) of the raw and treated water. Table (7) illustrates the interdependence of the raw water. Table (8), which illustrates the interdependence of the treated water. A positive correlation significant can be seen among some of the physical and chemical characteristics, but the high value of a positive correlation of raw water can be seen between Ca and Cl(R=0.8510. A positive correlation significant of treated water can be seen between Cl and all of So4, TSS, (R = 0.71), (R =
International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
SPSS statistical program was used for the purpose of conducting statistical analysis of the characteristics of the raw and treated water, and found a positive correlation significantly among
Tables (7) and (8) show the matrix of correlation(R) of the raw and treated water. Table (7) illustrates the interdependence of the raw
ositive correlation significant can be seen among some of the physical and chemical characteristics, but the high value of a positive correlation of raw water can be seen between Ca and Cl(R=0.8510. A positive
be seen between Cl and all of So4, TSS, (R = 0.71), (R =

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
51
Table (7): correlation matrix for raw water
Tem Tur. PH EC ALK T.H Ca Mg Cl So4 TDS TSS No3
Tem
Pearson Correlation
Sig. (2-tailed)
N
1
7
-.440
0.323
7
-.533
0.218
7
0.332
0.467
7
-.552
0.199
7
-.553
0.198
7
0.672
0.099
7
0.665
0.103
7
0.543
0.208
7
0.419
0.349
7
0.123
0.793
7
-.274
0.553
7
-.351
0.44
7
Tur.
Pearson Correlation
Sig. (2-tailed)
N
1
7
-.296
0.52
7
-.192
0.679
7
0.503
0.249
7
0.344
0.451
7
-.213
0.646
7
-.626
0.132
7
-.424
0.343
7
-.357
0.432
7
0.27
0.558
7
-.065
0.889
7
-.156
0.739
7
PH
Pearson Correlation
Sig. (2-tailed)
N
1
7
0.337
0.46
7
0.437
0.327
7
-.170
0.716
7
-.244
0.598
7
0.124
0.791
7
-.165
0.723
7
-.500
0.253
7
-.817*
0.025
7
0.648
0.115
7
-.103
0.826
7
EC
Pearson Correlation
Sig. (2-tailed)
N
1
7
0.17
0.716
7
-.713
0.072
7
0.381
0.398
7
0.693
0.084
7
-.033
0.943
7
-.583
0.17
7
-.600
0.154
7
0.277
0.548
7
-.857*
0.014
7
ALK
Pearson Correlation
Sig. (2-tailed)
N
1
7
0.122
0.795
7
-.161
0.731
7
-.301
0.512
7
-.322
0.481
7
-.372
0.412
7
-.374
0.409
7
0.423
0.345
7
-.219
0.637
7
T.H
Pearson Correlation
Sig. (2-tailed)
N
1
7
-.834*
0.02
7
-.507
0.245
7
-.633
0.127
7
0.145
0.756
7
0.507
0.245
7
-.579
0.173
7
0.659
0.108
7
Ca
Pearson Correlation
Sig. (2-tailed)
N
1
7
0.216
0.642
7
.851*
0.015
7
0.306
0.505
7
-.036
0.939
7
0.429
0.337
7
-.362
0.425
7
Mg
Pearson Correlation
Sig. (2-tailed)
N
1
7
0.048
0.918
7
-.137
0.77
7
-.468
0.289
7
-.173
0.711
7
-.573
0.179
7
Cl
Pearson Correlation
Sig. (2-tailed)
N
1
7
0.613
0.143
7
0.021
0.964
7
0.457
0.302
7
0.075
0.874
7
So4
Pearson Correlation
Sig. (2-tailed)
N
1
7
0.556
0.195
7
-.222
0.633
7
0.628
0.131
7
TDS
Pearson Correlation
Sig. (2-tailed)
N
1
7
-.614
0.142
7
0.56
0.191
7
TSS
Pearson Correlation
Sig. (2-tailed)
N
1
7
-.193
0.678
7
No3
Pearson Correlation
Sig. (2-tailed)
N
1
7

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
52
Table (8): matrix correlation for treated water
Correlations
Tem Tur. PH EC ALK T.H Ca Mg Cl So4 TDS TSS No3
Tem
Pearson Correlation
Sig. (2-tailed)
N
1
7
-0.511
0.241
7.000
-0.220
0.636
7.000
0.329
0.472
7.000
-0.468
0.290
7.000
-0.345
0.449
7.000
0.662
0.105
7.000
0.710
0.074
7.000
0.475
0.281
7.000
0.536
0.215
7.000
0.198
0.670
7.000
0.176
0.706
7.000
-0.307
0.503
7.000
Tur
Pearson Correlation
Sig. (2-tailed)
N
1.000
0.631
0.129
7.000
0.337
0.460
7.000
0.072
0.879
7.000
0.044
0.925
7.000
-0.476
0.280
7.000
0.123
0.794
7.000
-0.566
0.185
7.000
-0.928
0.003
7.000
-0.553
0.198
7.000
-0.279
0.545
7.000
-0.334
0.464
7.000
PH
Pearson Correlation
Sig. (2-tailed)
N
1.000
0.512
0.240
7.000
0.278
0.546
7.000
-0.528
0.223
7.000
-0.483
0.272
7.000
0.479
0.277
7.000
-0.699
0.080
7.000
-0.721
0.068
7.000
-0.649
0.115
7.000
-0.435
0.329
7.000
-0.561
0.190
7.000
EC
Pearson Correlation
Sig. (2-tailed)
N
1.000
0.231
0.619
7.000
-0.685
0.090
7.000
0.406
0.366
7.000
0.691
0.085
7.000
-0.125
0.789
7.000
-0.471
0.286
7.000
-0.581
0.172
7.000
-0.063
0.894
7.000
-0.792
0.034
7.000
ALK
Pearson Correlation
Sig. (2-tailed)
N
1.000
-0.105
0.822
7.000
-0.229
0.621
7.000
-0.332
0.466
7.000
-0.587
0.166
7.000
-0.379
0.401
7.000
0.013
0.978
7.000
-0.521
0.231
7.000
-0.190
0.682
7.000
T.H
Pearson Correlation
Sig. (2-tailed)
N
1.000
-0.262
0.570
7.000
-0.674
0.097
7.000
-0.061
0.896
7.000
0.033
0.943
7.000
0.594
0.160
7.000
-0.257
0.578
7.000
0.353
0.437
7.000
Ca
Pearson Correlation
Sig. (2-tailed)
N
1.000
0.261
0.572
7.000
0.776
0.040
7.000
0.427
0.339
7.000
-0.064
0.892
7.000
0.541
0.210
7.000
-0.257
0.578
7.000
Mg
Pearson Correlation
Sig. (2-tailed)
N
1.000
-0.006
0.990
7.000
-0.087
0.853
7.000
-0.342
0.453
7.000
-0.019
0.967
7.000
-0.595
0.159
7.000
Cl
Pearson Correlation
Sig. (2-tailed)
N
1.000
0.701
0.079
7.000
0.029
0.951
7.000
0.874
0.010
7.000
0.306
0.504
7.000
So4
Pearson Correlation
Sig. (2-tailed)
N
1.000
0.568
0.184
7.000
0.466
0.292
7.000
0.534
0.217
7.000
TDS
Pearson Correlation
Sig. (2-tailed)
N
1.000
-0.286
0.534
7.000
0.454
0.306
7.000
TSS
Pearson Correlation
Sig. (2-tailed)
N
1.000
0.422
0.346
7.000
No3
Pearson Correlation
Sig. (2-tailed)
N
1.000

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
53
7-RESULTS
1- Sedimentation efficiency (65.97%) is considered acceptable, with what must be done by a sedimentation phase during the detention time of the sedimentation basins.
2- Filtration efficiency was ranged between 59% in January 2014 and 75% in the month of December 2013 needs to be developed by following the washing and ongoing maintenance of the filters periodically.
3- The efficiency of the plant in terms of biological and special treatment E-coli bacteria is not good and volatile, especially during the month of November 2013 and March and April 2014, and this poses a threat to the health of consumers. The bodies of non-technical was lifting bottles of chlorine gas was a great danger to the lives of citizens and exposing them to disease.
4- Physical and chemical properties of treated water were equipped to the specifications of the World Health Organization (WHO) and raw water is characterized good quality properties.
5- It was noted that the security authorities prevented the use of chlorine gas sterilization for the period from early 2014 until mid-April of the same year, in spite of the importance of the need for sterilization.
8-RECOMMENDATIONS
1- Environmental awareness must be increasing among the workers at the station for an alert to the
risk of contamination and how to treat and emphasize the need for non-interference by any outside parties; and under any justification in the operation and management of the station, to the threat this poses to the lives of citizens and exposing them to disease.
2- Physical, chemical and biological properties need to conduct water and supplying scientific laboratory qualified specialist, especially in the field of bio-examination.
3- Filters were washing and maintenance of the regularly and continuously to maintain the qualitative characteristics of water processed for citizen so as to ensure continued compliance with the international standard specifications WHO.
4- The plant was need for regular maintenance and correcting the imbalance and cleaning sedimentation basins and clarification on a regular basis and scheduler.
5- Tests must be done for some of the other elements that are not measured in this study such as fluoride because of this element of the impact on the dental health of consumers, especially children.
9-REFERENCES
[1] Allaa M. Aenab, S. K. Singh "Evaluation of Drinking Water Pollution and Health Effects in
Baghdad, Iraq", IVSL.org Journal of Environmental Protection, No 3, 2012, pp 533-537. [2] Ahmed AL-Sarawy, (basic processes for drinking water purifications) Book 1st edition,
scientific book house for diffusion and distribution, Cairo2011. (in Arabic language). [3] Goel, P.K, (water pollution causes effect and control) Book Published by New age
International, New Delhi, 1997. [4] PNW Water (history of drinking water) Use and treatment Pacific Northwest Regional water
program, 2008. [5] Awny Ahmed Mohamed, (the water from the source to the landfill) Environment Public
Authority, Tripoli – Libya,(in Arabic Language), 2002. [6] Shaheen, Khalid Mohammed. “Evaluation study of a water treatment plant in the left side of
the city of Mosul (Quppa project)”, Technical Journal. (in Arabic Language), Volume 17, No. 3.2004

International Journal of Civil Engineering and Technology (IJCIET), ISSN 0976 – 6308 (Print),
ISSN 0976 – 6316(Online), Volume 5, Issue 6, June (2014), pp. 37-54 © IAEME
54
[7] Abbas A. Al-Jeebory, Dr .Ali H. Ghawi, “Performance Evaluation of AL-Dewanyia water treatment plant in Iraq”, Al-Qadisiya Journal For Engineering Sciences Vol. 2 No.4 ,2009.
[8] Mohammed, Ali Ahmed, alaa-A, Shakir, “Evaluation the performance of al-wahdda project Drinking water treatment plant”, A case study in Iraq, international journal of advances in applied sciences (IJAAS), Vol.1, No.3, 2012 September, pp.130-138.
[9] Directorate of Kirkuk Water Management ( hawija water treatment plant)charts and data. (in Arabic language), 2007.
[10] APHA, (standards methods for the examination of water and wastewater) 18th ed. (1992). [11] Steel .EW and Mcghee.T.J (water supply& sewerages) 5th edition, 1979. [12] Al-Layla, M.A., Ahmanded, Sh., Middle brooks, E.J., (Design of Water Supply Engineering),
Ann Arbor Science Publisher, INC. (in Arabic Language), 1977. [13] Majeed Mattar Ramal, "Evaluating the Drinking Water quality supplied by the Large
Treatment Plant in RAMADY City", Al-Qadissiya Journal of Engineering, Vol.3, No.2, 2010.
[14] Abdul Rah man, Ibrahim Abdul Karim and Mawllod I., Ahmed Saud, Oran Menem, "Evaluate the quality of drinking water and efficiency of Fallujah water Treatment plant", Iraqi Journal of Civil Engineering,.(In Arabic Language), Volume VI, Issue I, June 2004
[15] AL-Ganema, A.A.R., Bacteriological Quality for the water of AL-Remedy governorate, MSc thesis, College of Science, Anbar University, 1995.
[16] World Health Organization (WHO), (guidelines for drinking water quality-Health Criteria
and other supporting information) 2ndEdition, Vol .2 Geneva, 1996. [17] Abdulmuhsin S. Shihab, Waleed M.Sh. Al-Abidrabah and Ahmad Kh. Ibrahim, “Statistical
Analysis of Groundwater Quality Parameters in Selected Sites at Ninavah Governorate/ Iraq”, International Journal of Civil Engineering & Technology (IJCIET), Volume 5, Issue 4, 2014, pp. 57 - 70, ISSN Print: 0976 – 6308, ISSN Online: 0976 – 6316.
[18] R Radhakrishanan and A Praveenkar, “Sustainability Perceptions on Wastewater Treatment Operations in Urban Areas of Developing World”, International Journal of Civil Engineering & Technology (IJCIET), Volume 3, Issue 1, 2012, pp. 45 - 61, ISSN Print: 0976 – 6308, ISSN Online: 0976 – 6316.
[19] Neeraj D. Sharma and Dr. J. N. Patel, “Experimental Study of Groundwater Quality Improvement by Recharging with Rainwater”, International Journal of Civil Engineering & Technology (IJCIET), Volume 2, Issue 1, 2011, pp. 10 - 16, ISSN Print: 0976 – 6308, ISSN Online: 0976 – 6316.
[20] R. S. Sapkal and Dr. S. S. Valunjkar, “Development and Sensitivity Analysis of Water Quality Index for Evaluation of Surface Water for Drinking Purpose”, International Journal of Civil Engineering & Technology (IJCIET), Volume 4, Issue 4, 2013, pp. 119 - 134, ISSN Print: 0976 – 6308, ISSN Online: 0976 – 6316.










![[XLS] · Web view1 2 2 2 3 2 4 2 5 2 6 2 7 2 8 2 9 2 10 2 11 2 12 2 13 2 14 2 15 2 16 2 17 2 18 2 19 2 20 2 21 2 22 2 23 2 24 2 25 2 26 2 27 2 28 2 29 2 30 2 31 2 32 2 33 2 34 2 35](https://static.fdocuments.in/doc/165x107/5aa4dcf07f8b9a1d728c67ae/xls-view1-2-2-2-3-2-4-2-5-2-6-2-7-2-8-2-9-2-10-2-11-2-12-2-13-2-14-2-15-2-16-2.jpg)







![content.alfred.com · B 4fr C#m 4fr G#m 4fr E 6fr D#sus4 6fr D# q = 121 Synth. Bass arr. for Guitar [B] 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 5](https://static.fdocuments.in/doc/165x107/5e81a9850b29a074de117025/b-4fr-cm-4fr-gm-4fr-e-6fr-dsus4-6fr-d-q-121-synth-bass-arr-for-guitar-b.jpg)