1/21/2013 NO SCHOOL - MLK DAY. 1/22/2013 Standards: 3 (Stoichiometry) Objectives: ● be able to:...
-
Upload
douglas-stafford -
Category
Documents
-
view
214 -
download
0
Transcript of 1/21/2013 NO SCHOOL - MLK DAY. 1/22/2013 Standards: 3 (Stoichiometry) Objectives: ● be able to:...

1/21/2013
NO SCHOOL - MLK DAY

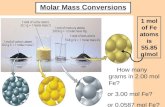
1/22/2013 Standards: 3 (Stoichiometry)Objectives:● be able to: mol <---> mass (compounds)DO NOW: 1. Convert 4.100 mol KMnO4 to grams of KMnO4
(check ans = 647.8 g). If correct, start homework.HOMEWORK: 1. Ch 11.3, p. 323 - 324 27-29, and 30 (a-e). Show all calculations. All
problems require 2 fractions, including given/1. Check you work, answer will be on today's HW page. (5 pts) Std 3.
==========================================================BASKET: Journal 1/18 (Molar Mass Lab) 1. Ch 11.3 p. 321 20-24, and p. 322 25-26 (5 pts) Std 3

When done, turn over ans sheet and quiz




DO NOW
Convert 4.100 mol KMnO4 to grams of KMnO4


1/23/2013 Standards: 3 (Stoichiometry)Objectives:● be able to: mass <---> mol <---> particles (compounds)DO NOW: 1. Convert 85.0 g KMnO4 to particles of KMnO4 . Requires 3 fractions
including the given/1.HOMEWORK: 1. Ch 11.3, p. 326 31-35. Show all calculations and label each section of each problem. (5 pts) Std 3==========================================================BASKET: 1. Ch 11.3, p. 326 31-35. Show all calculations. All problems
require 3 fractions, including given/1. (5 pts) Std 3

ANNOUNCEMENTLook at your on-line grade and your Stamp Sheet and compare to my
on-line stamp sheet. If you have anything missing, you have until Friday to make-up or receive a note home to parents.


DO NOW
1. Convert 85.0 g KMnO4 to particles of KMnO4

p. 326 #31
Particles in this problem are:
Ag1+
Ag1+
IN SOLUTION
Ag
Ag
Cr
O
O
O
O
CrO4
2- ION
Ag2CrO4 THE SOLID
ZERO CHARGElook below
look below
Cr
O
O
O
O
Demo - AgNO3 mix with NaCl

p. 326 #31
first convert 25.8 g Ag2CrO4 to particles (molecules Ag2CrO4 ). You cannot answer a, b, and c until you have done this conversion.
Solution:
a. convert particles of Ag2CrO4 to particles of Ag1+ ions.
b. convert particles of Ag2CrO4 to particles of CrO4
2- ions.
c. Finding the grams of one Ag2CrO4 molecule means the grams/formula unit of Ag2CrO4 . That is the molar mass of Ag2CrO4 divided by the Avo number.
ratio of Ag2CrO4 : Ag1+ ions is: 2 Ag1+ : 1 Ag2CrO4 Use this ratio to convert Ag2CrO4 particles (answer to first part of solution)
25.8 g Ag2CrO4 1 mol Ag2CrO4 6.02 x 1023 Ag2CrO4
----------------------- x ---------------------- x -----------------------------1 332 g Ag2CrO4 1 mol Ag2CrO4
= 0.468 x 1023 = 4.68 x 1022 Ag2CrO4
ratio of Ag2CrO4 : CrO4
2- ions is: 1 CrO4
2- : 1 Ag2CrO4 Use this ratio to convert Ag2CrO4 molecules (answer to first part of solution) to CrO4
2- ions
Ag
AgC
r
OO OO
332 g Ag2CrO4 1 mol Ag2CrO4
1 mol Ag2CrO4 6.02 x 1023 Ag2CrO4
x 5.51 x 10-22 g/formula unitAg2CrO4
=
4.68 x 1022 Ag2CrO4 2 Ag1+
1 1 Ag2CrO4
x = 9.36 Ag1+ ions
4.68 x 1022 Ag2CrO4 1 CrO4
2-
1 1 Ag2CrO4
x = 4.68 X 1022 CrO4
2- ions

1/24/2013 Standards: 3 (Stoichiometry)Objectives:● be able to calculate: mass <---> mol <---> particles (compounds)DO NOW: 1. Complete Worksheet on mass <---> mol <---> particles HOMEWORK: 1. App A, p. 876-877, Ch 11.3 15-24 (5 pts) Std 3==========================================================BASKET (Turned In): 1. Ch 11.3, p. 326 31-35. Show all calculations and label each section of each problem. (5 pts) Std 3
ANNOUNCEMENTSPractice quiz (10 pts) on Friday 1/25 and
Periods 1 & 2 notebooks to be collected Friday 1/25.

Answers to tonight's HW so you can check your answers.
Ch 11.3, App A, p. 876 15-24

Notebooks collected for Periods 1 & 2 Fri 1/25 Periods 3 Mon 1/28 Periods 5 & 6 Tu 1/29
1/25/2013 Standards: 3 (Stoichiometry)Objectives:● Review: mass <---> mol <---> particles (compounds)● Mini-Quiz Ch 11.1-11.3 (10 pts)DO NOW: 1. Turn in HW and take a blank piece of paper.HOMEWORK: 1. App A, p. 876-877, Ch 11.3 15-24 (5 pts) Std 3 (Yes, DO IT AGAIN)
If you do not have a text book, you will find the problems on the next page of this Whiteboard file.
==========================================================BASKET (Turned In): Journal 1/23 (2 days ago) 1. App A, p. 876-877, Ch 11.3 15-24 (5 pts) Std 3
Students were ill prepared for this quiz.

App A, p. 876-877, Ch 11.3 15-24 (5 pts) Std 3



FI ND
GIV EN
2 students - one move arrows, one dictates set-upGiven Find




Formula of a Hydrate
moles of H2O
moles of dry salt

HYDRATES
Chemicals that adsorb water.
Dried out hydrates will get "wet" if left out in an unsealed container.
They will even get "wet" in a "closed" container if left long enough.

Example of hydrate Calculations
BaCl2 • 2 H2O
Getting the moles of H2O
Mass of evaporating dish = 15.9 gMass of dish and hydrated salt = 400.0
Mass of hydrated salt 400.0 gMass of dry salt 368