1 Quiz on _________ Single Daily Grade. 3 Table of Contents Difference between Molecular and Ionic...
-
Upload
georgiana-barton -
Category
Documents
-
view
243 -
download
0
Transcript of 1 Quiz on _________ Single Daily Grade. 3 Table of Contents Difference between Molecular and Ionic...


4

5
A. Definition
•Remember, a compound consists of 2 or more elements chemically bonded together. It is always neutral.
EX: H2O, NaCl, Sb3(PO4)5

6
•There are two main ways to name chemical compounds. However, before you can even think about naming the compound you must decide what kind of compound you have. Do you have a covalent or ionic compound? We already learned this using the terms molecule and formula unit.

7
•Most formulas are written with the elements listed from left to right on the periodic table

8
What do you remember about the difference between a molecule and a formula unit?

B. Types of Compounds
2 types of chemical compounds

10
SamplesIonic
Compounds• Rock salt (NaCl)• Calcium Carbonate
(CaCO3)
• Potassium Nitrate (KNO3 )
Molecular Compounds
• Sugar (C12H22O11)
• Methane gas (CH4)
What’s the difference?

11
1. Ionic Compound:Ionic Compound: a compound that is
formed by ions of opposite charges. It is held together by an ionic (transfer electrons) bond and usually contains a metal and nonmetal.
EX: NaCl, Sb3(PO4)5
smallest unit is called a formula unit

12
2. Molecular or Covalent Compound:
• Molecular or Covalent compound: consists of only nonmetal atoms and is held together by covalent (share electrons) bonds
EX: H2O, C8H18
smallest unit is called a molecule

13
How can I tell!?
•Look for a metal.
•If there’s a metal, it’s an ionic compound

14
Practice1. Determine whether the
following are ionic or molecular covalent compounds
a. N2O5 b. PbNO5
c. KF d. AgCl
e. PCl3
ionic
molecular
molecular
ionic
ionic
KF AgCl
PbNO5

15
Acids: have a formula that starts with hydrogen
You need to know the names and formulas for the following acids:
Acid Name Formula
hydrochloric acid HCl
nitric acid HNO3
sulfuric acid H2SO4
phosphoric acid H3PO4
Note: all acids start with “H” in their chemical formulas and include the word
“acid” in their names.

II. Molecular Compounds
(aka Covalent Compounds aka
Molecules)

17
Molecular Compounds = share electrons
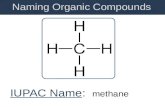
H O
H2OH

18
A. Binary Molecular Compounds
•Molecular compounds DO NOT contain a metal atom
•Molecular compound contain covalent bonds
•We use prefixes to name molecular, ONLY MOLECULAR, compounds

19
How do I distinguish between the bikes?
unicycle bicycle tricycle

20
We use prefixes to help distinguish molecular
compounds.Prefix Number of Atoms
mono 1
di 2
tri 3
tetra 4
penta 5
hexa 6
hepta 7
octa 8
nona 9
deca 10

21
Why have prefixes?Nitrogen bonds to Oxygen
in more than one way
Ex: N2O, NO2, N2O2, N2O5

22
Examples:Formula: CO
Name:
Formula: N2O
Name:
carbon monoxide
dinitrogen monoxide

23
a. Naming binary molecular compounds:
FIRST ELEMENT IN THE FORMULA – keeps its full elemental name – Add prefixes to signify the number of
that specific kind of atom ONLY if there is more than one of the atoms. DON’T USE MONO ON THE FIRST SYMBOL
SECOND ELEMENT IN THE FORMULA– The second element name ALWAYS
gets a prefix– Keep the first syllable of the
elemental name and change the suffix to “ide”.

24
Practice:Formula: CO2 name:Formula: BF3 name:Formula: Cl2O name:Formula: P2S5 name:
Carbon dioxide
Boron trifluorideDichlorine monoxideDiphosphorus pentasulfide

25
B. Converting a name to a formula:
•This exercise is very simple if you have memorized the element symbols and the prefixes. Just go backwards from what we just did.

26
Examples:Formula: Name: carbon tetrachloride.
Formula: Name: dihydrogen monofluoride
CCl4
H2F

27
Practice:Formula: H2O name: Dihydrogen monoxideFormula: name: Nitrogen pentoxide Formula: name: CarbonTetrafluoride Formula: name: Silicon Dioxide
NO5
CF4
SiO2

1+
2+ 3+ 3- 2- 1-

29
III. Naming ions1. Monatomic Cations (+ ions)
•Monatomic cations are named by writing the element’s name and then “ion” after it.
•EX: Ca2+ Na+
One atom +
Sodium ion
Calcium ion

30
a. Monatomic Cations (Transition + PASS + Z.A.C
Elements )• Most transition metals and “P.A.S.S” elements need Roman Numerals
• Zinc (Zn), Silver (Ag), and Cadmium (Cd) are transition metals that DO NOT need Roman Numerals “Z.A.C Elements”
• Roman Numerals 1-5: I, II, III, IV, V

31
More than just “Iron ion”
EX: Fe2+ Fe3+
Iron (III) ionIron (II) ion

32
Transition Metals need R.N.1. Name these
ionsa. Pb4+ b. Pb2+
c. Cu+ d. Cu2+
Lead (IV) ionLead (II) ionCopper (I) ionCopper (II) ion

33
Examples1. Name these ionsa. Ca2+ b. Na+
c. Fe4+ d. Zn2+
Calcium ionSodium ionIron (IV) ionZinc ion

34
b. Monoatomic Anions (- ions)
oMonatomic anions are named by changing the suffix to “ide” then adding “ion” after it.

35
Cation or Anion?• Carbon can act as a cation (carbon
ion) or as an anion (carbide ion). It just depends if it is in the beginning or the end of the compound’s formula
• Hydrogen can also act as a cation (hydrogen) or as an anion (hydride). It just depends if it is in the beginning or the end of the compound’s formula. Don’t forget it is a non-metal.

Complete the chart below
Element SymbolOxidation number
Anion name
Chlorine Cl- -1 Chloride ion
Bromine Br- -1 Bromide ion
Oxygen
S2-
Phosphide ion
N3-
Sulfur
O2- -2 Oxide ion
-2 Sulfide ion
Phosphorus P3- -3
Nitrogen -3 Nitride ion

37
Summary•List the elements that need Roman Numerals
•List the transition metals that don’t need Roman Numerals
Transition metals, PASS elements (Pb, As, Sn, Sb)
Zn, Ag, Cd (Z.A.C elements)

38
Always need roman numerals
1+
2+ 3+ 3- 2- 1-
TipsTM (Except ZAC) and PASS need Roman NumeralsAll metals are +; all non-metals are -Anions end in -ide
Anions: Change Suffix to -ide
Zn2+ Cd2+Ag+
always

39
c. Polyatomic Cations
•A polyatomic ion is a group of covalently bonded atoms that have an overall charge
•Example NH4
+ = ammonium.

40
d. Polyatomic Anions • This means the anion is a
COVALENTLY BONDED GROUP of atoms that have a – charge. They are EASY to deal with in naming and formula writing if you think of them as ONE UNIT that can not be altered in any way.

41
Example:
•Use the back of your periodic table to help you identify polyatomic anions

42
Flip over your PT and let’s practice
1. Name these ionsa. CO3
2- b. OH- c. NO3
- d. SO4
2-
e. PO43-
carbonatehydroxidenitratesulfatephosphate

44
IV. Binary Ionic Compoundsa. Naming Binary Ionic Compounds
• Ionic Compounds are made of a cation and anion pair. You already know how to name the ions. Just put the names together and leave off the word ion. Don’t forget Roman Numerals!

45
Ionic Compounds = transfer of electrons
Na Cl+ -
NaClIs formed

46
a. Naming Binary Ionic Compounds
Formula
Ions Ion names Compound name
AlNAl3+
N3- Aluminum ionNitride ion
Na20Na+
O2-
Sodium ionOxide ion
FeOFe2+
O2-
Fe2O3Fe3+
O2-
Iron (II) ionOxide ion
Iron (III) ionOxide ion
Aluminum nitride
Sodium oxide
Iron (II) oxide
Iron (III) oxide

47
Ex. Name FeS
Iron Sulfide
Iron (II) sulfide
NOT

48
Remember to add a Roman Numeral for the cations that need one
One trick to determine the Roman Numeral:1. Add up your total negative
charges2. Divide the above number by the
total number of metal ions3. Change to a Roman
Numeral

49
Ex. Name FeS
Total neg. charges: 2-2/1 (Fe) = 2
1. Add up your total negative charges2. Divide the above number by the
total number of metal ions3. Change to a Roman
Numeral
Iron (II) Sulfide

50
Ex. Name Fe2O3
Total neg. charges: 6-6/2 (Fe) = 3
Iron (III) Oxide

51
Practice
1. Name the following compounds
a. ZnS
b. K3N
c. BaO
d. CaBr2
e. SnF2
Zinc sulfide
Potassium nitride
Barium oxide
Calcium bromide
Tin (II) fluoride

52
Practice
1. Name the following compounds
f. CaO
g. AlF3
h. CuI2
i. Cu2Se
j. Hg2O
Calcium oxide
Aluminum fluoride
Copper (II) iodide
Copper (I) SelenideMercury (I) Oxide

53
b. Writing Formulas for Binary Ionic Compounds
•Remember, a compound must be electrically neutral. Therefore, the total positive charge of the cation must equal the total negative charge of the anion.

54
EX: calcium chloride
Ca2+ Cl-
To make a neutral compound Ca2+ Cl-
Cl-+2 -2
CaCl2

55
To write a formula for a binary ionic compound1.Write the ions that are named in the compound showing oxidation numbers.2. Determine how many of each ion you need to make a neutral compound.3. To write the formula, list the metal symbol first and then the anion symbol, adding subscripts to indicate how many of each ion there is in the formula4. Make sure the formula has the lowest whole number ratio.
Note: you never write a 1 subscript; it is understood.

56
EX: write the formula for aluminum fluoride
Al+3 F-
NOTE: You never include oxidation numbers in the chemical formula of a compound.
Formula: AlF3
F-
F-
+3 -3

57
Practice – only do a-c1. Write chemical formulas for the following:
a. strontium phosphide
b. potassium sulfide
c. iron (II) chloride
Sr3P2
K2S
FeCl2

58
The Criss-Cross Method
•One easy way to figure out the formula of an ionic compound is to swap the oxidation number of each ion and use them as subscripts. (without the charge)
•the formula must be in the lowest whole number ratio

59
Na S
Just Watch!!
sodium sulfide
1 2+ -
1’s are understood
Na2S

60
Fe Br
Ex: Write the formula for iron (III) bromide
3+ -
FeBr3

61
Mg S
Ex: Write the formula for magnesium sulfide
2 2+ -
Note: if both ox #’s are equal, you don’t need to use the
crisscross method, just show 1 ion of each.

62
Magnesium Sulfide
MgS(not Mg2S2)

63
Pb S
Note: show the formula with the lowest whole number ratio.
EX: write the formula for lead (IV) sulfide
4 2+ -

64
Criss Cross Method
Pb2S4
PbS2

65
Finish the Practice Box1. Write chemical formulas for the following:
d. antimony (V) nitride
e. iron (III) chloride
f. Calcium bromideCaBr2
FeCl3
Sb3N5

66
Naming
•Same way as you already learned
•Use subscripts to show how many of each ion you needed to make a neutral compound
Formulas

67
V. Ionic Compounds Containing Polyatomic
Ions a. Writing Formulas for Polyatomic Compounds
• Write formulas for polyatomic ions in the same way. Just remember the polyatomic ion acts as a whole unit and you need to use parentheses if you need more than 1 of them.
• Note: You never change the • formula of a polyatomic ion!

68
EX: calcium nitrate
To make a neutral compound Ca2+
(NO3)-
(NO3)-
+2 -2Ca(NO3)2

69
Ca (NO3)
EX: Calcium Nitrate(using the criss cross
method)
2+ -

70
PracticePractice1. Write formulas for the following
compoundsa. strontium sulfate
b. lithium carbonate
c. potassium sulfate
d. magnesium hydroxide
SrSO4 or Sr(SO4)
Li2CO3 or Li2(CO3)
Mg(OH)2
K2SO4 or K2(SO4)

71
PracticePractice1. Write formulas for the following
compoundse. ammonium dichromate
f. potassium permanganate
g. sodium hypochlorite
h. barium sulfite
(NH4)2Cr2O7 or (NH4)2(Cr2O7)
NaClO or Na(ClO)
BaSO3 or Ba(SO3)
KMnO4 or K(MnO4)

72
b. Naming Polyatomic Compounds
•It is important that you recognize the polyatomic
• If there are more than 2 elements in the formula, then you know it contains a polyatomic.
•Only one polyatomic is a cation, ammonium (NH4
+) All the rest will come after the metal ion.

73
•You name polyatomic compounds just like you would other ionic compounds -- you write the name of each ion. Note: most (not all) polyatomic compounds will end in -ate or -ite.

74
Ex. Name ZnSO4
Zinc
Ex. Name NaOH
Sodium
Sulfate
hydroxide

75
Practice1. Name these compoundsa. CaCO3
b. KClO
c. KMnO4
calcium carbonate
potassium hypochlorite
potassium permanganate

76
Practice1. Name these compounds
d. Al(OH)3
e. Sn3(PO4)2
f. Na2CrO4
aluminum hydroxide
tin (II) phosphate
sodium chromate