目录 - ie.tsinghua.edu.cn · fishbone. above. As our experiments are restricted to many...
-
Upload
phamnguyet -
Category
Documents
-
view
212 -
download
0
Transcript of 目录 - ie.tsinghua.edu.cn · fishbone. above. As our experiments are restricted to many...
1 / 20
Cooling rate of water Group 5: Xihui Yuan, Wenjing Song, Ming Zhong,
Kaiyue Chen, Yue Zhao, Xiangxie Li
目录 1. Abstract: .............................................................................................................................. 1
2. Introduction: ...................................................................................................................... 2
2.1. Statement of the problem: ................................................................................... 2
2.2 objectives: ................................................................................................................. 2
2.3. Rationale .................................................................................................................. 2
3. Literature review ............................................................................................................... 2
3.1. Basic Concept .......................................................................................................... 3
3.2 Existing experiment on the cooling rate of water ........................................... 3
3.3.Predictive conclusions ........................................................................................... 4
4. Methods ............................................................................................................................... 4
4.1. Experiment variables ............................................................................................. 4
4.3. Design of experiment ............................................................................................ 7
4.4. Instrument ............................................................................................................... 8
4.5. Performing the experiment .................................................................................. 9
5. Analysis ............................................................................................................................. 10
5.1. Data analysis ......................................................................................................... 10
5.2. Conclusion ............................................................................................................. 18
6. In-depth research ............................................................................................................ 19
7. Future extension ............................................................................................................. 19
8. Reference .......................................................................................................................... 20
1. Abstract:
This thesis aims at exploring factors that can affect the cooling rate of water.
Water is the foundation of life; however, although we are very familiar with it, it
still has many aspects that we do not quite understand. There are some existing
theories and experiments about this topic, on this basis, we will do further study
to find cooling rate under different conditions. In order to explore the effectors
that have an influence on the cooling condition, we find out different variables,
and select proper ones to be considered in our experiment according to the scope
of our research. Some 2 level factors, factorial designs will be undertaken, and
several analytical methods will be applied to the data got from experiments, until
we get to the ultimate goal and get acquaintance of the cooling procedure.
2 / 20
2. Introduction:
2.1. Statement of the problem:
Water is one of the most common things in our daily life. Nearly all the
students take a bottle of water or hot drink with them every day. Naturally, how to
keep their drink warm in the winter and make hot drink cool as quick as possible
becomes a question that many will concern. But at the same time, few of them
have put their thoughts into action. As a result, this area of knowledge for most
people remains to be relatively blank.
2.2 objectives:
In this experiment, we will measure the cooling rate of different drinks in
different environment, to draw one or more conclusions about better ways of
keeping drink warm in winter and cooling water quickly in summer.
2.3. Rationale
Our experiment is focused on cooling rate of different drinks. As cooling rate
will be relatively certain and stable under certain conditions, so the data will be
meaningful and we can certainly draw some conclusions from it. Different factors
of environment conditions, holding bottles and initial temperature will be
considered. The cooling rate will be measured by thermometer, guaranteeing the
correctness and accuracy of the experiments.
This experiment is focused on water or drink’s cooling in our daily life, so the
experiment subjects will be the most commonly and easily found drink in our life.
And the variables of experiment environment will be designed within the
limitation of real life as well.
3. Literature review
To get some predictive conclusions before the experiment is a very important
method to give us some intuition to estimate whether the experiment is
performing correctly and whether there are some significant wrong message in
the final result. In order to get some intuition and predictive conclusion before the
experiment, we find some literature related to our experiment and do some
forecasts before we do the experiment.
3 / 20
3.1. Basic Concept
There are some very important formulas related closely to our cooling rate
experiment.
Heat conduction formular: 𝑄 = 𝑐𝑚∆𝑇
𝑄 = 𝑡ℎ𝑒 𝑞𝑢𝑎𝑛𝑡𝑖𝑡𝑦 𝑜𝑓 ℎ𝑒𝑎𝑡 𝑐 = 𝑡ℎ𝑒 𝑠𝑝𝑒𝑐𝑖𝑓𝑖𝑐 ℎ𝑒𝑎𝑡 𝑐𝑎𝑝𝑎𝑐𝑖𝑡𝑦 𝑚 = 𝑡ℎ𝑒 𝑞𝑢𝑎𝑛𝑡𝑖𝑡𝑦 𝑜𝑓 𝑡ℎ𝑒 𝑙𝑖𝑞𝑢𝑖𝑑 ∆𝑇 = 𝑡ℎ𝑒 𝑑𝑖𝑓𝑓𝑒𝑟𝑒𝑛𝑐𝑒 𝑏𝑒𝑡𝑤𝑒𝑒𝑛 𝑡ℎ𝑒 𝑖𝑛𝑖𝑡𝑖𝑎𝑙 𝑡𝑒𝑚𝑝𝑒𝑟𝑎𝑡𝑢𝑟𝑒 𝑎𝑛𝑑 𝑡ℎ𝑒 𝑓𝑖𝑛𝑎𝑙 𝑡𝑒𝑚𝑝𝑒𝑟𝑎𝑡𝑢𝑟𝑒
Newton's law of cooling 𝑄 = 𝑞 × 𝑠 × 𝑡
𝑞 = 𝛾 ×𝑇0 − 𝑇1
𝑑
𝛾 = 𝑎 𝑐𝑜𝑛𝑠𝑡𝑎𝑛𝑡 𝑟𝑒𝑙𝑎𝑡𝑒𝑑 𝑡𝑜 𝑡ℎ𝑒 𝑡𝑒𝑥𝑡𝑢𝑟𝑒 𝑜𝑓 𝑡ℎ𝑒 𝑐𝑜𝑛𝑡𝑎𝑖𝑛𝑒𝑟 𝑇0 = 𝑡ℎ𝑒 𝑖𝑛𝑖𝑡𝑖𝑎𝑙 𝑡𝑒𝑚𝑝𝑒𝑟𝑎𝑡𝑢𝑟𝑒 𝑜𝑓 𝑡ℎ𝑒 𝑙𝑖𝑞𝑢𝑖𝑑
𝑇1 = 𝑡ℎ𝑒 𝑡𝑒𝑚𝑝𝑒𝑟𝑎𝑡𝑢𝑟𝑒 𝑜𝑓 𝑡ℎ𝑒 𝑒𝑛𝑣𝑖𝑟𝑜𝑛𝑚𝑒𝑛𝑡 𝑑 = 𝑡ℎ𝑒 𝑡ℎ𝑖𝑐𝑘𝑛𝑒𝑠𝑠 𝑜𝑓 𝑡ℎ𝑒 𝑐𝑜𝑛𝑡𝑎𝑖𝑛𝑒𝑟 𝑠 = 𝑡ℎ𝑒 𝑎𝑟𝑒𝑎 𝑜𝑓 𝑡ℎ𝑒 𝑖𝑛𝑡𝑒𝑟𝑓𝑎𝑐𝑒 𝑡 = 𝑡𝑖𝑚𝑒 𝑜𝑓 𝑡ℎ𝑒 𝑐𝑜𝑜𝑙𝑖𝑛𝑔 𝑒𝑥𝑝𝑒𝑟𝑖𝑚𝑒𝑛𝑡
After combining all the formulas above, we can have the following formula:
∆𝑇 = 𝛾 ×𝑇0 − 𝑇1
𝑑× 𝑆 ×
𝑡𝑐𝑚
3.2 Existing experiment on the cooling rate of water
Some experiment show that cooling rate has the relationship with the initial
temperature of the water. When the volume is constant, the relationship between
freezing time and initial temperature has the following curve as Graph 1:
4 / 20
After finding some materials about this topic, we find that the cooling rate of
water may have the relationship with the initial temperature, the volumes, the
shape of the container, the material of the container and ambient temperature etc.
3.3.Predictive conclusions
1. If the other factors are treated as constants, the thicker the container is (d↑), the smaller ∆𝑇 will be.
2. If the other factors are treated as constants, then the greater the mass is (m↑), the smaller ∆𝑇 will be.
3. If the other factors are treated as constants, then the larger of the specific
heat capacity, the smaller ∆𝑇 will be.
4. If the other factors are treated as constants, then the higher the initial
temperature is , the larger the ∆𝑇 will be.
4. Methods
4.1. Experiment variables
The key dependent variable of our experiment is the cooling rate of water,
there are many aspects can influent it.
Initial temperature
The tim
e to
freeze (
it
)
Graph 1
5 / 20
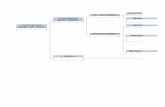
Graph 2 _ fishbone There are mainly three aspects affecting the cooling rate of water: intrinsic
factors of water, environment influences, and anthropic factors. Within each of
these topics, there are still many detail factors; these are reflected in the Graph 2 _
fishbone above.
As our experiments are restricted to many conditions, some of the variables
we can’t control, and other of them are not within our scope of study, so we
explore further the influence of all variables to our experiment and give them
detail analysis.
6 / 20
Table 1
response
variable
(units)
normal
operating level
& range
meas. precision,
accuracy
relationship of
response variable to
objective
Cooling rate 5-10 centigrade
in ten minutes
Thermometer
Precision 0.01
centigrade
Estimate the cooling
condition of
different water
under different
conditions
Table 2
control
variable
(units)
normal level
& range
meas.
precision
& setting error
proposed settings,
based on
predicted effects
predicted
effects
Cooling time
(in minutes) 5-10min
in ten seconds
watch 5 minutes different
Initial
temperature
(1 centigrade)
30-100
centigrade thermometer
Range from
30-100 centigrade
according to
different
experiment
different
solution Water vs.
milk select
Same brand, “Yili”
Milk different
thickness of
the
containers
( in ml)
1-2 layers of
paper cup count
1-2 layers of paper
cup according to
different
experiments
different
Volume of
the liquid 0-200ml
Measuring
cylinder 50-150ml different
Table 3
fixed
factor
(units)
desired
experimental
level &
allowable
range
measurement
precision
How known?
how to
control in experiment
anticipated
effects
Cooling
time
5 minutes
Within 10s
0.1s
stopwatch
Cool water of in
different trails in 5
None to
slight
7 / 20
minutes, time controlled
by stopwatch
Table 4
nuisance
factor (units)
measurement
precision
strategy (e.g.,
randomization,
Blocking, etc.)
anticipated effects
Variation of
condition of
environment
In 1centigrade
By thermometer
Randomization
experiment
sequence
none
Evaporation amount
In 0.01ml, by
measuring
cylinder
Measure
volume after
heating
slight
Difference in paper
cups In o.1 millimeter Eliminate None to slight
Air velocity 0.1-0.3 m/s
Close door and
window, stay
away from the
cups whiling
cooling
Too much air flow
can cause large
influence
4.3. Design of experiment
Factors related to the cooling rate of water are based on comparisons among
series of experiments. The comparison in this experiment is basically in two
dimensions, the first dimension focus on the cooling rate of different liquid, we
would like to find out the cooling speed of some certain kind of liquid, for
example, pure water, milk; further more in this dimension, we will compare the
differences in cooling rate and use tools to find the related factors to cooling
speed. Then we will move forward to design the experiments aiming at finding the
relation and the function of these factors.
The second dimension aims at finding the factors related to the environment,
for example the temperature and the thickness of the cup.
The design method we choose is 2K full factorial design. The response variable
is cooling degree in 5 minutes; the factors are initial temperature, volume,
thickness of cups, and liquid type. Each factor has two levels, (60 centigrade vs. 80
centigrade), (50ml vs. 100ml), (1layer vs. 2layers), (water vs. milk)
Factor Range Actual level Coded level Type
Initial
temperature 60℃-80℃
60℃
80℃
-1
+1 Quantitative
Quantity of 50ml-100 ml 50 ml -1 Quantitative
8 / 20
liquid 100 ml +1
Thickness of
container 1-2 layers
1 layer (cup)
2layers(cups)
-1
+1 Quantitative
Type of liquid
Water
Milk
-1
+1 Qualitative
Use Minitab to generate a 24 full factorial design with two replications.
Graph 3
4.4. Instrument
Pure water: solvent and one kind of the experimental liquid.
Tap water, milk of the same brand (Yili)
Sanitary cup: hold different kinds of liquid, in the same material.
Alcohol thermometer: monitor the change and show the temperature.
One 100ml beaker and one 150ml beaker: get the desired amount of the
experimental liquid.
9 / 20
Air thermometer: get the environment temperature and humidity.
4.5. Performing the experiment
In order to improve the efficiency of the experiment, we did some trials to
find proper values for the parameters and ranges.
Considering the availability, samples of 50ml, 60℃ water were used to
choose the time parameter; the experiment was started at the point when the
temperature is 60℃ and after 3min and 5min separately, the ending temperature
was recorded. The difference for 3min was 3℃, and 8℃ for 5 min. Considering
the error of measurement and the significance of the data, 5min was finally
chosen to be the time parameter.
The staring temperature was also adjusted. It was originally desired to set the
to be 70 ℃ and 90 ℃, in the trial experiment, 90℃ was found hard to control,
because the procedure of using the beaker to measure the proper volume and
then pouring into the cups could always lead to a fall below 90℃, and with the
limitation of the instrument, the original temperature of water in the boiler was
not essentially higher than 90℃. With more trials of monitoring the temperature
of water in the boiler and in the cups after the transporting, the range of
temperature was set to be 60℃ and 80℃.
Then, the formal experiment was conducted, first, the temperature of
environment was recorded to be 30.2℃, and the humidity was 23%.
The experiment was carried out according to the randomized sequence. A
beaker was used to take the desired volume of water out from the boiler, then the
water was poured into the numbered cups and the thermometer was used to
monitor the temperature until it fell down to the starting point. Then, start the
timer and wait for 5 minutes, record the ending temperature and do the next one.
As to the milk, it was heated also in the boiler, within its bag. The heated milk
was poured into the beaker to get the desired volume, in order to slow down the
cooling speed in measuring; the beaker was also heated in the water boiler. After
the measurement, the milk was poured into the cup; the thermometer was used to
monitor the temperature until it fell down to the starting point. Then, start the
timer and wait for 5 minutes, record the ending temperature and do the next one.
Perform all the items in the table and record the temperature of the ending
point.
In the whole process, as the experiment was frequently switched between the
water and milk, two beakers were used to measure the volume, and the
instrument used should keep clean. Because the temperature could change very
quickly around the desired points, great attention was paid on monitoring the
temperature.
After the experiment, the temperature of the environment was 31℃. And the
humidity was 24%. This tiny change was not taken into consideration as factors in
the experiment.
10 / 20
5. Analysis
5.1. Data analysis
Step 1: calculate the cooling degree.
The initial table records the starting temperature and the finishing
temperature, so we first calculate the difference of these two columns and get the
cooling degree.
Step 2: normal verification
First of all, we need to check the weather the response variable is normal
distributed. So we do the normal test and find that under 0.05 significant level the
response variable “温差” is not normal distributed(Graph 4). Then we do the
Box-Cox transformation (Graph 5 Box-Cox transformation). We find λ = 0, so we
do the Ln transformation (Graph 6 Normal test of Ln 温差).
11 / 20
Graph 4
Graph 5 Box-Cox transformation
3020100-10
99
95
90
80
70
60
50
40
30
20
10
5
1
温差
百分
比均值 13.5
标准差 6.048
N 32
AD 0.779
P 值 0.039
温差 的概率图正态 - 95% 置信区间
5.02.50.0-2.5-5.0
50
40
30
20
10
Lambda
标准
差
CL 下限 CL 上限
限
估计 0.10
CL 下限 -0.67
CL 上限 1.01
取整值 0.00
(使用 95.0% 置信)
Lambda
温差 的 Box-Cox 图
12 / 20
Graph 6 Normal test of Ln 温差
Step 3: find significant factors and their influences
The main effect plot(Graph 7 Main effect plot) and the interaction plot(Graph
8 Interaction plot of the factors) show that the factors 起温, 水量, 杯厚 and the
interaction of 液体*水量 , 液体*杯厚 seem to have significant effect on the
response variable. More specifically, when the initial temperature rises, the
cooling degree increases, the volume of liquid decreases, the cooling degree
increases, and increase the thickness of cup, may result in the decline of cooling
degree. What’s more, from the main effect plot, we can find that the type of liquid
has no significant influence on the cooling rate.
4.03.53.02.52.01.51.0
99
95
90
80
70
60
50
40
30
20
10
5
1
ln温差
百分
比
均值 2.497
标准差 0.4804
N 32
AD 0.571
P 值 0.127
ln温差 的概率图正态 - 95% 置信区间
13 / 20
Graph 7 Main effect plot
Graph 8 Interaction plot of the factors
And we also draw the contour plot (Graph 9) and surface plot(Graph 10) to
show the relationship of the factors and response variable. From the plots, we can
see that the factors mainly have liner relationship with the response variable.
牛奶水
2.8
2.6
2.4
2.2
2.08060
10050
2.8
2.6
2.4
2.2
2.021
液体
平均
值
起温
水量 杯厚
ln温差 主效应图数据平均值
8060 21
3.0
2.5
2.0
3.0
2.5
2.0
3.0
2.5
2.0
牛奶水
3.0
2.5
2.0
10050
液体
起温
水量
杯厚
水
牛奶
液体
60
80
起温
50
100
水量
1
2
杯厚
ln温差 交互作用图数据平均值
14 / 20
起温*液体
10-1
1.0
0.5
0.0
-0.5
-1.0
水量*液体
10-1
1.0
0.5
0.0
-0.5
-1.0
杯厚*液体
10-1
1.0
0.5
0.0
-0.5
-1.0
水量*起温
10-1
1.0
0.5
0.0
-0.5
-1.0
杯厚*起温
10-1
1.0
0.5
0.0
-0.5
-1.0
杯厚*水量
10-1
1.0
0.5
0.0
-0.5
-1.0
液体 -1
起温 -1
水量 -1
杯厚 -1
保持值
>
–
–
–
–
–
–
< 2.0
2.0 2.2
2.2 2.4
2.4 2.6
2.6 2.8
2.8 3.0
3.0 3.2
3.2
ln温差
ln温差 的等值线图
Graph 9
12.4
0
2.7
3.0
-1
3.3
0 -11
l n 温差
起温
液体
12.0
0
2.2
2.4
-10 -1
1
l n 温差
水量
液体
1
2.2 0
2.3
2.4
-1
2.5
0 -11
l n 温差
杯厚
液体
1
2.00
2.5
-1
3.0
0 -11
l n 温差
水量
起温
1
2.1 0
2.4
2.7
-1
3.0
0 -11
l n 温差
杯厚
起温
12.0
0
2.1
-1
2.2
0 -11
l n 温差
杯厚
水量
液体 -1
起温 -1
水量 -1
杯厚 -1
保持值
ln温差 的曲面图
Graph 10
15 / 20
Step 4: explore relationship function
The normal probability plot of effect (Figure 8) and the Pareto plot (Figure 9)
have showed the same result that we draw from the main effect plot and
interaction plot.
2520151050-5-10
99
95
90
80
70
60
50
40
30
20
10
5
1
标准化效应
百分
比
A 液体
B 起温
C 水量
D 杯厚
因子 名称
不显著
显著
效应类型
ABD
AD
AC
D
C
B
标准化效应的正态图(响应为 ln温差,Alpha = .05)
Graph 11
A
BCD
ACD
BC
ABCD
AB
ABC
BD
CD
ABD
D
AD
AC
C
B
2520151050
项
标准化效应
2.12
A 液体
B 起温
C 水量
D 杯厚
因子 名称
标准化效应的 Pareto 图(响应为 ln温差,Alpha = .05)
Graph 12
16 / 20
We assume that the high order terms are not significant. And we use only the
one and two order significant factors to do the regression, the final result is as
below:
2520151050-5-10
99
95
90
80
70
60
50
40
30
20
10
5
1
标准化效应
百分
比
A 液体
B 起温
C 水量
D 杯厚
因子 名称
不显著
显著
效应类型
AD
AC
D
C
B
标准化效应的正态图(响应为 ln温差,Alpha = .05)
Graph 13
A
D
AD
AC
C
B
20151050
项
标准化效应
2.06
A 液体
B 起温
C 水量
D 杯厚
因子 名称
标准化效应的 Pareto 图(响应为 ln温差,Alpha = .05)
Graph 14
17 / 20
Table 5 一般线性模型: ln 温差 与 液体, 起温, 水量, 杯厚 因子 类型 水平数 值
液体 固定 2 水, 牛奶
起温 固定 2 60, 80
水量 固定 2 50, 100
杯厚 固定 2 1, 2
ln 温差 的方差分析,在检验中使用调整的 SS
来源 自由度 Seq SS Adj SS Adj MS F P
液体 1 0.0003 0.0003 0.0003 0.03 0.866
起温 1 5.3127 5.3127 5.3127 451.33 0.000
水量 1 1.1749 1.1749 1.1749 99.81 0.000
杯厚 1 0.1169 0.1169 0.1169 9.93 0.004
液体*水量 1 0.1359 0.1359 0.1359 11.54 0.002
液体*杯厚 1 0.1182 0.1182 0.1182 10.04 0.004
误差 25 0.2943 0.2943 0.0118
合计 31 7.1531
S = 0.108495 R-Sq = 95.89% R-Sq(调整) = 94.90%
项 系数 系数标准误 T P
常量 2.49710 0.01918 130.20 0.000
液体
水 -0.00326 0.01918 -0.17 0.866
起温
60 -0.40746 0.01918 -21.24 0.000
水量
50 0.19162 0.01918 9.99 0.000
杯厚
1 0.06044 0.01918 3.15 0.004
液体*水量
水 50 -0.06516 0.01918 -3.40 0.002
液体*杯厚
水 1 -0.06077 0.01918 -3.17 0.004
The fitted model is:
ln 温差 = 2.4971 − 0.40746I起温=60 + 0.19162I水量=50 + 0.06044I杯厚=1
− 0.06516I液体=水,水量=50 − 0.06077I液体=水,杯厚=1
So
18 / 20
温差 = EXP(2.4971 − 0.40746I起温=60 + 0.19162I水量=50 + 0.06044I杯厚=1
− 0.06516I液体=水,水量=50 − 0.06077I液体=水,杯厚=1)
Step 5: residual analysis:
The residual plot (Graph 15 Residual plot) is satisfied. We find that there is an
outlier, but we don’t know the reason now. Maybe it is the result of reading error.
And it is a full factorial design; it may cause unbalance if we delete this point.
Then we keep it in the plot.
Graph 15 Residual plot
5.2. Conclusion
1. From the equation 温差 = EXP(2.4971 − 0.40746I起温=60 + 0.19162I水量=50 +
0.06044I杯厚=1 − 0.06516I液体=水,水量=50 − 0.06077I液体=水,杯厚=1) and analysis of
data, we find that high initial temperature, less water yield and thin cup cause
large cooling rate. It obeys our common sense.
2. Whether the liquid is milk and water does not affect the cooling rate. We only
test these two liquid, so we do not sure whether this conclusion can be extend
to other liquid.
0.20.0-0.2-0.4
99
90
50
10
1
残差
百分
比
3.53.02.52.01.5
0.1
0.0
-0.1
-0.2
-0.3
拟合值
残差
0.10.0-0.1-0.2-0.3
8
6
4
2
0
残差
频率
3230282624222018161412108642
0.1
0.0
-0.1
-0.2
-0.3
观测值顺序
残差
正态概率图 与拟合值
直方图 与顺序
ln温差 残差图
19 / 20
6. In-depth research
In our model, the type of liquid is not a significant factor, however, we want to
know whether this is the truth or some other elements influence the result of
experiment.
After referring to some literatures, we find the heat formulae can calculate the
difference of degrees in our experiment conditions
According to Q=CM△T
Where C 水=4.2×10^3J/(Kg℃)
C 牛奶=2.5×10^3J/(Kg℃)
ρ水= 1.0X10^3kg/m^3
ρ牛奶= 1.0256~1.0288 X10^3kg/m^3
Get the △T ≈2 ℃
We compute the difference of cooling centigrade under same experiments for
both water and milk, and make a single sample T test, get
Table 6 差
异
-2 -2 -1 -1 -1 -1 -1 -1 0 0 0 0 1 1 2 2
Table 7 单样本 T: 差异
mu = 0 与 ≠ 0 的检验
平均值
变量 N 平均值 标准差 标准误 95% 置信区间 T P
差异 16 -0.250 1.238 0.310 (-0.910, 0.410) -0.81 0.432
According the test result, the difference incurred by type of liquid cannot be
identified by the measuring system, so in this experiment, the liquid type is not
significant.
7. Future extension
Enhance the evaluation system:
The resolution of the alcohol thermometer is 1 centigrade, this is not precise
for our experiment, so in order to enhance the accuracy of the experiment, we
20 / 20
need to improve the measuring system.
Extend the experiment parameter range:
Extend the experiment range: choose more types of liquid and extend the range of
initial water, we may get more precise conclusions about cooling rate of different
liquid. Also, under this condition, the factors increase, we need to do fractional
factorial design or Taguchi design to explore the relationships.
8. Reference
http://www.eqna.org/15541/how-does-dissolving-sugar-affect-the-cooling-rate-o
f-water
《水、盐水降温试验及分析计算》刘永胜,杨小林,谢华刚 (河南理工大学土木建筑系,河南焦
作 454003)




































![Beer Fishbone Diagram - Rotated[1]](https://static.fdocuments.in/doc/165x107/55400001550346a57f8b493e/beer-fishbone-diagram-rotated1.jpg)

